At NIMBE (UMR CEA, CNRS, Université Paris-Saclay), researchers from the LIONS laboratory developed an automated method to simulate the chemical kinetics of complex gas mixtures exposed to ionizing radiation. In radiation chemistry, the main challenge is not only energy deposition but the rapid multiplication of transient species (ions, radicals, excited states), which leads to large, highly coupled reaction networks. Traditionally, such networks were built manually, restricting modeling efforts to relatively simple or highly purified systems.
Small modular reactors (SMRs) are increasingly considered as low-carbon electricity sources supporting electrification strategies. In conventional schemes, nuclear energy is converted into heat, then electricity, and eventually chemical energy. An alternative pathway, first explored in the mid-20th century, consists in directly converting part of the reactor’s ionizing radiation into chemical energy – the so-called chemonuclear reactor concept.
Because SMRs have higher surface-to-volume ratios, the fraction of “loss radiation” (radiation not converted into usable heat and absorbed in shielding) can increase, which may be harnessed for chemistry without major core design changes. The key question is which chemical transformations can be effectively driven by such radiation, and under what conditions.
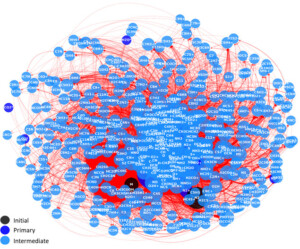
To address this issue, the LIONS team developed a nonselective automated method for generating radiolysis reaction networks based on curated open-access kinetic databases (KIDA, UMIST, GRIMECH) and solved using the FACSIMILE software. This approach overcomes a major methodological limitation: modeling complex gas mixtures where numerous transient species generate thousands of coupled reactions.
The framework was first validated on a well-documented CO₂/O₂/CO reference system, successfully reproducing experimental trends in a low-to-moderate conversion regime (≈10–20%). It was then applied to a representative flue gas mixture (N₂/CO₂/H₂O/O₂/CO/NO₂/SO₂), leading to networks comprising up to 448 species and 4,780 reactions. A parametric study (pressure, temperature, additives) revealed promising composition domains: CO₂ conversion increases fivefold with 90 vol.% NH₃, and adding H₂S can yield hydrogen production exceeding 900 molecules·100 eV⁻¹ under specific conditions.
Overall, the study shows how systematic simulation can identify “radiocatalytic” regimes worthy of experimental investigation, providing a rigorous framework for rapid screening of nuclear-to-chemical concepts and for exploring innovative pathways for industrial gas valorization (including CO₂) in the energy transition context.
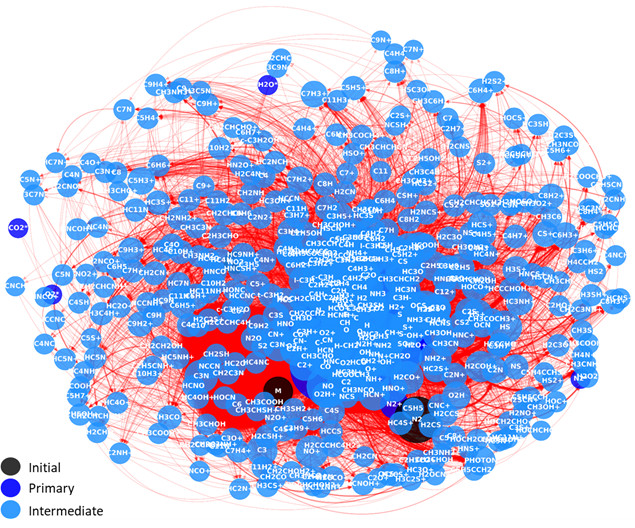
Fig. Graph network representing the system of elementary reactions under study for the simulation of flue gas under ionising radiation. Each node is a chemical specie. Each arrow represents a reaction from a reactant to a product.
Reference
« A Nonselective Automatic Method for Kinetic Simulations of Gas Mixtures under Ionizing Radiation: Case Study of a Nuclear-to-Chemical Concept”, Antoine Dion, Marie Géléoc, Jean-Philippe Renault, Chemistry-Methods, 2025.