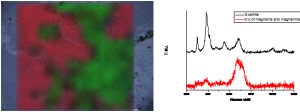
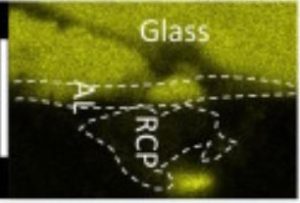
The Raman microspectrometry is an analytical device of primary importance to characterize the crystalline structure of materials. It is a complementary technique to micro X-ray diffraction. This technique is well suited to characterize and determine the distribution of the phases formed in the corrosion layers of iron or steels during very long periods in various environments. Iron oxides, oxy-hydroxides and carbonates are the main phases encountered in these systems. At the LAPA thick corrosion layers of artefacts corroded during several ten or hundred years are examined thanks to Raman microspectroscopy. The aim of these characterizations is to determine the distribution of the phases formed in the corrosion layers on samples prepared on transverse sections.
The LAPA is specialized in the understanding of the long term corrosion of iron in various environments. Iron oxides, oxy-hydroxides and carbonates are the main phases encountered in these systems. They are studied on transverse sections of archaeological artefacts. The aim of these characterizations is to determine the distribution of these phases in the corrosion layers. Mapping acquisitions are well suited to answer to these questions (Figure). The exploitation of these data is conducted following different methods with the help of the Wire software developed by Renishaw. The first one is based on the Region of Interest (Signal to baseline methods). But the dataset can also be exploited by supervised or not supervised multi-variate analyses.
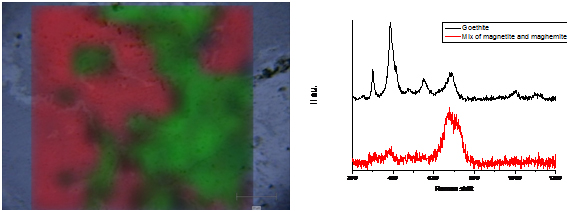
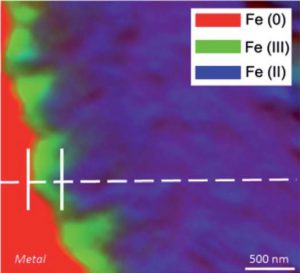
Different experiments allow also to understand the corrosion mechanisms. For example experiments of recorrosion are conducted in a D2O solution in order to identify the localization of the precipitation of the corrosion phases on archaeological artefacts. The identification of the deuterated carbonated phases and their localization at the interface between the corrosion products and metal by Raman spectroscopy conducts to describe the long term corrosion mechanisms of iron in an anoxic environment. The electrochemical behavior of the phases present inside the corrosion layers has been explored thanks to an electrochemical cell set up under the Raman microspectrometer. For example maghemite put under a cathodic potential is reduced in magnetite and reoxidised into maghemite when aerated conditions are reconducted.
The Raman spectrometer of the LAPA is an Invia reflex from the Renishaw company. It is equipped with a frequency-doubled Nd: YAG emitting at 532 nm and a solid state laser emitting at 473 nm. The laser beam is focused on the sample through an optical microscope and a x50, long working distance x50 and a x100 LEICA objectives. The laser spot size on the samples is less than 2 µm. A 2400 l/mm grating induces a spectral resolution of about 1 cm-1. The detector is a charge-coupled device
multichannel matrix detector operating at −70 ◦C (by air cooling). The laser beam power on the surface of the sample can be modulate through optical density filters. Thanks to a motorized stage mapping acquisitions can be collected. The hyperspectral images obtained provides fruitful information on the localization of various crystalline phases constituting the observed systems.
Neff, D., L. Bellot-Gurlet, et al. (2006). « Raman imaging of ancient rust scales on archaeological iron artefacts for long-term atmospheric corrosion mechanisms study. » Journal of Raman Spectroscopy 37(10): 1228-1237.
Neff, D., S. Reguer, et al. (2004). « Structural characterization of corrosion products on archaeological iron. An integrated analytical approach to establish corrosion forms. » Journal of Raman Spectroscopy 35: 739-745.
Reguer, S., D. Neff, et al. (2007). « Deterioration of iron archaeological artefacts: micro-Raman investigation on Cl-containing corrosion products. » Journal of Raman Spectroscopy 38(4): 389-397.
Saheb, M., D. Neff, et al. (2010). « Raman study of a deuterated iron hydroxycarbonate to assess long-term corrosion mechanisms in anoxic soils. » Journal of Raman Spectroscopy: n/a-n/a.