In collaboration with chemists of ENS Lyon and biophysicists of DSV (IBiTeC-S), a team of LSDRM (IRAMIS/SIS2M) designed a cellular probe taking advantage of the extreme sensitivity of NMR (Nuclear Magnetic Resonance) of hyperpolarized xenon*. The experiment shows the excellent selectivity of the method to detect the chosen molecular target . This work paves the way to an ultra sensitive molecular MRI (Magnetic Resonance Imaging) that can be used in vivo and allowing early detection of extra-intracellular biological events.
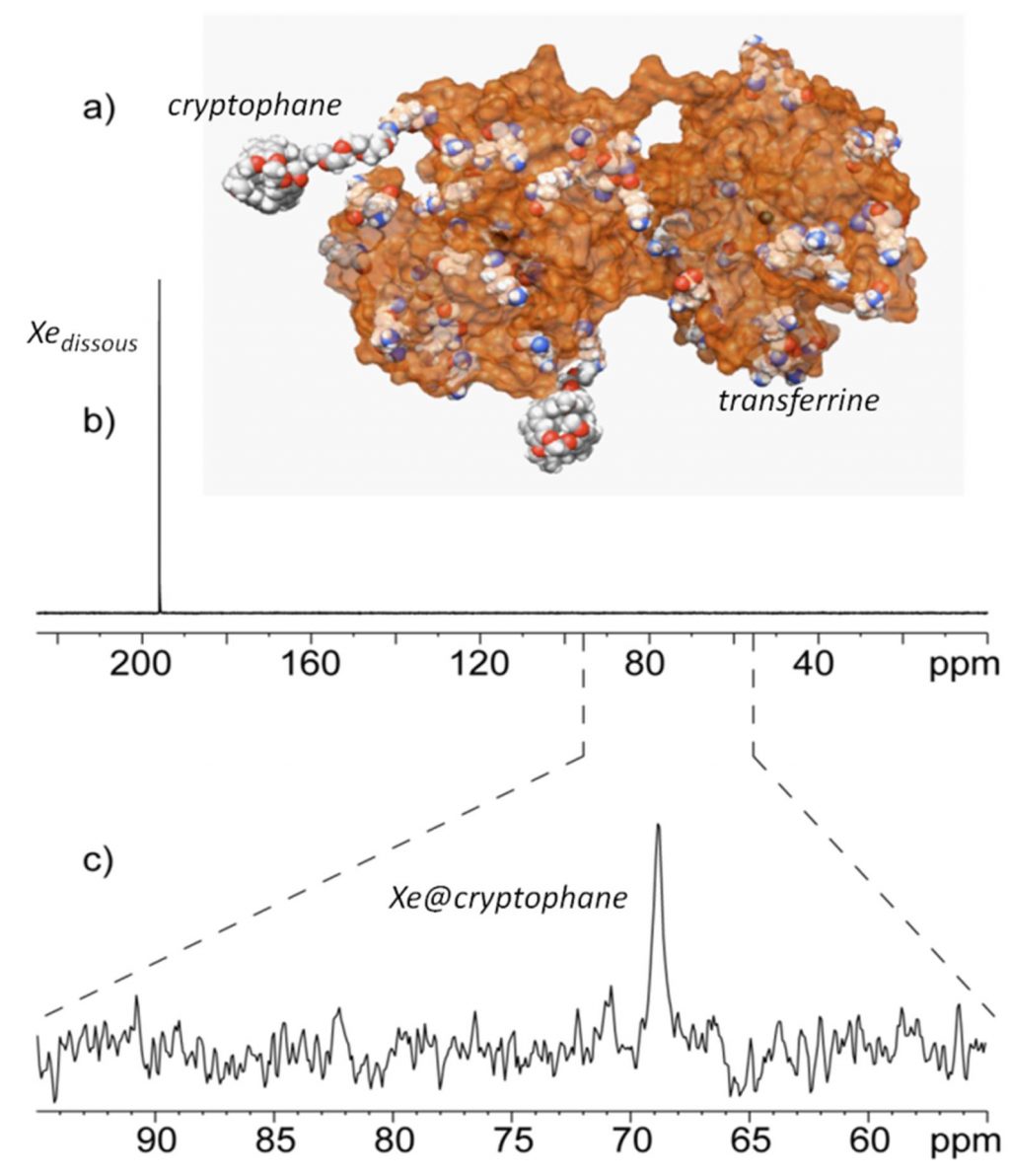
The targeted system is the transferrin whose role is to internalize iron into cells. The idea was to design a biosensor by grafting cryptophanes, host molecules of xenon, on the protein of 80 kDa. The use of polarized xenon allows a gain in NMR detection sensitivity of about 104 (Figure 1a). So encapsulated in cryptophanes, xenon has its own spectral signature,allowing to distinguish it from isolated Xe. Associated with the use of laser-polarized xenon, this provides a probe:
- specific
- identifiable
- and detectable with very high sensitivity (Figure 1c).
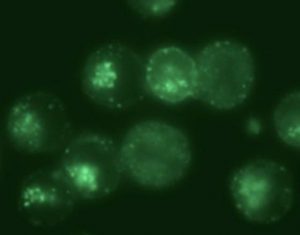
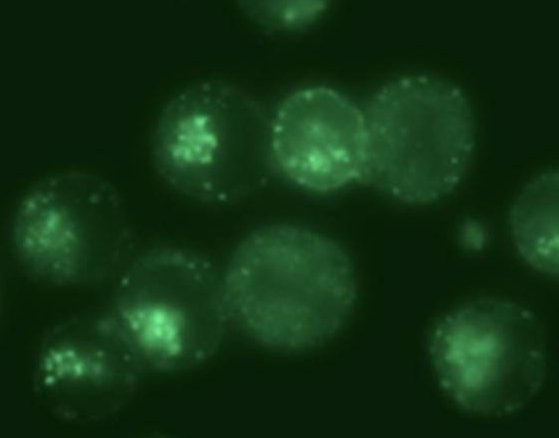
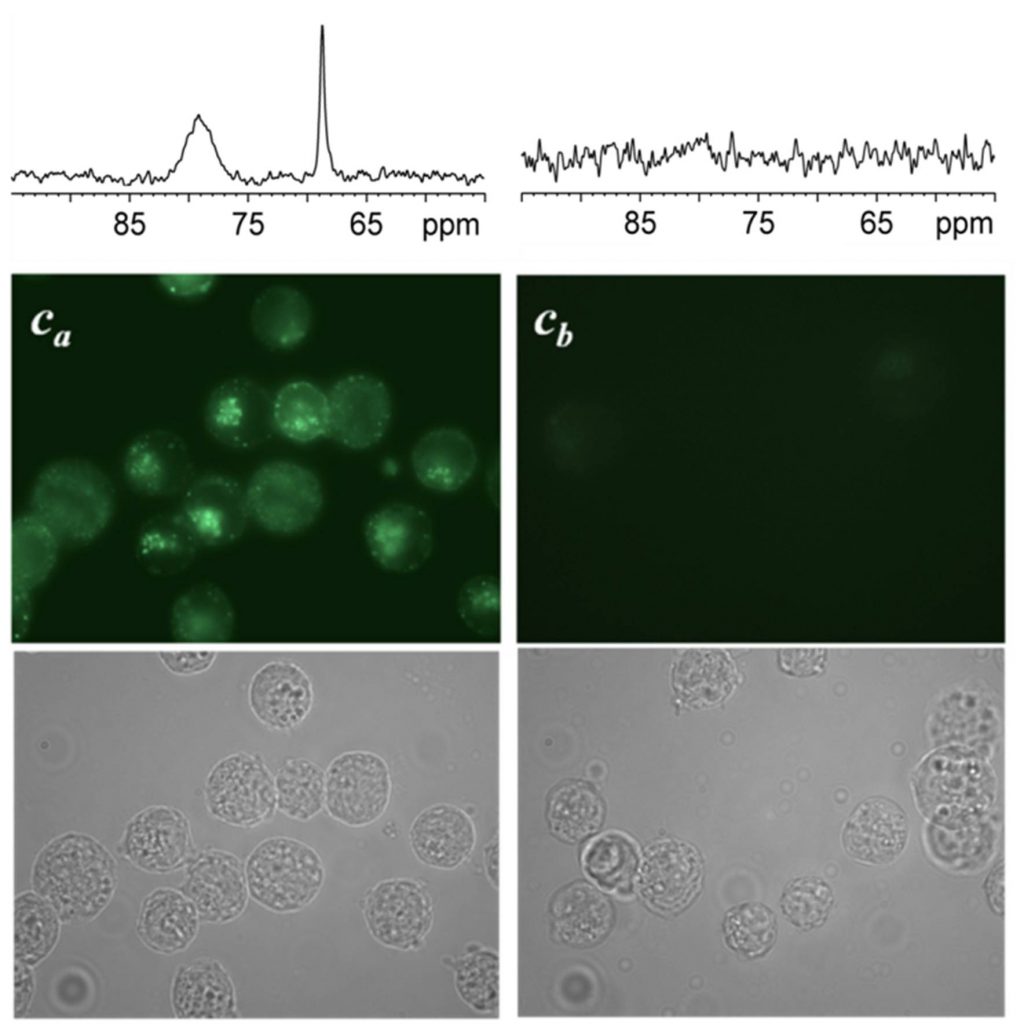
When eukaryotic cells expressing the transferrin receptors are incubated with this biosensor, the NMR spectrum reveals, after rinsing and elimination of the portion of unfixed biosensors, not one but two signals in the spectral region of encapsulated xenon (Figure 2, Ca cells). They correspond respectively to biosensors in a membrane environment (signal at 79 ppm) and in an aqueous environment (69 ppm). This spectroscopic observation is entirely consistent with fluorescence images showing the cellular internalization of the biosensor, and also its presence within the plasma membrane.
A second batch of cells (Cb) was treated with pronase, enzyme destroying the surface receptor of transferrin, before being incubated with the bioprobe. In this case, always in agreement with the fluorescence images, no signal is displayed in the region of encapsulated xenon after rinsing of the cells. Although the cells are still upright, the suppression of the transferrin receptors prevents the internalization of bioprobe to take place.
This shows that the bioprobe is well specific to the transferrin receptors. By NMR of hyper-polarized xenon, it is possible to study its interaction with cells and in particular to detect its internalization.
This work opens the way for ultra-sensitive molecular MRI that can be used in vivo, allowing early detection of biological extra- and even intra-cellular events.
While previous bioprobes had allowed the detection of targets in isotropic solution (see for example “The polarized xenon for imaging“), detection by NMR of hyperpolarized xenon of intra-cellular biological phenomena has been achieved for the first time on a sample of living cells, demonstrating the in vivo applicability of the method.
* hyperpolarization: out of equilibrium state where populations of nuclear spins are strongly away from the Boltzmann distribution. For xenon, this state can be obtained by transfer order with photons (optical pumping).
References:
– Cell Uptake of a biosensor detected by hyperpolarized 129Xe NMR: the transferrin case
C. Boutin, A. Stopin,F. Lenda, T. Brotin, J.-P. Dutasta, N. Jamin, A; Sanson, Y. Boulard, F. Leteurtre, G. Huber, A. Bogaert-Buchmann, N. Tassali, H. Desvaux, M. Carrière and P. Berthault, in press in 'Bioorganic and Medicinal Chemistry’.
– Hyperpolarized 129Xe NMR signature of living biological cells
Céline Boutin,1 Hervé Desvaux,1 Marie Carrière,1 François Leteurtre,2 Nadège Jamin,2 Yves Boulard 2 and Patrick Berthault1, NMR in Biomedicine (2011)*.
– Effect of pH and counterions on the encapsulation properties of xenon in water-soluble cryptophanes
P. Berthault, H. Desvaux, T. Wendlinger, M. Gyejacquot, A. Stopin, T. Brotin, J.P. Dutasta, Y. Boulard, Chemistry. 16, (2010) 12941-12946.
– See also the highlights :
- – IRAMIS-IBiTec-S (Janvier 2011) : “Le xénon: un atome sensible à son environnement“.
- – IRAMIS (Mars 2009) : “Towards record levels of sensitivity for NMR by the measurement of spin-noise spectra' “
- – IRAMIS (Septembre 2008) : “Toward a new molecular imaging using hyperpolarized 129Xe NMR biosensors“
- – IRAMIS-IBiTec-S (2007) : “Le xénon polarisé pour l'imagerie“
[1] C. Boutin, H. Desvaux, M. Carrière, P. Berthault : CEA, IRAMIS, SIS2M, Laboratoire Structure et Dynamique par Résonance Magnétique, UMR CEA/CNRS 3299, F-91191 Gif-sur-Yvette (France)
[2] N. Jamin, F. Leteurtre, Y. Boulard : CEA, IBiTec-S/SBIGeM, F-91191 Gif-sur-Yvette (France)
[*] Support from the French Ministry of Research (project ANR-06-PCVI-0023 BULPOXI) is acknowledged.
Contact: P. Berthault.