Researchers from the LEDNA group at the NIMBE laboratory (CEA-CNRS) in collaboration with several international partners have demonstrated that the electronic structure of nanodiamonds can be engineered to generate solvated electrons under visible light illumination. This advance opens new avenues for photocatalysts capable of harnessing solar energy to drive highly demanding chemical reactions, such as the reduction of carbon dioxide (CO₂).
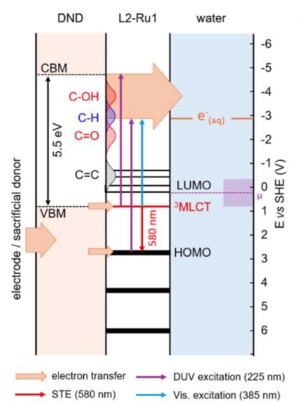
Solvated electrons are among the most powerful reducing species known in chemistry. In water, their reduction potential reaches approximately -2.9 V (vs NHE), enabling the activation of highly stable molecules such as carbon dioxide. Hydrogenated nanodiamond is a promising material for generating these electrons, as its surface exhibits a unique electronic structure that, upon illumination, enables the formation and solvation of electrons in water. However, a major limitation hinders these applications: the wide bandgap of diamond (~ 5.5 eV) requires deep ultraviolet light excitation, which is energetically expensive and may damage the diamond-solvant interfaces.
To overcome this limitation, the researchers developed a strategy to engineer electronic states by grafting a molecular photosensitizer derived from a ruthenium tris-bipyridine complex onto nanodiamonds. Anchored to the surface through tailored organic linkers, this molecule introduces new electronic levels within the diamond band gap. Advanced spectroscopic analyses, including X-ray absorption spectroscopy, ultraviolet photoelectron spectroscopy, and transient absorption spectroscopy, reveal a strong interaction between the dye and the nanodiamond, whichcan drive the emission of solvated electrons under visible light illumination.
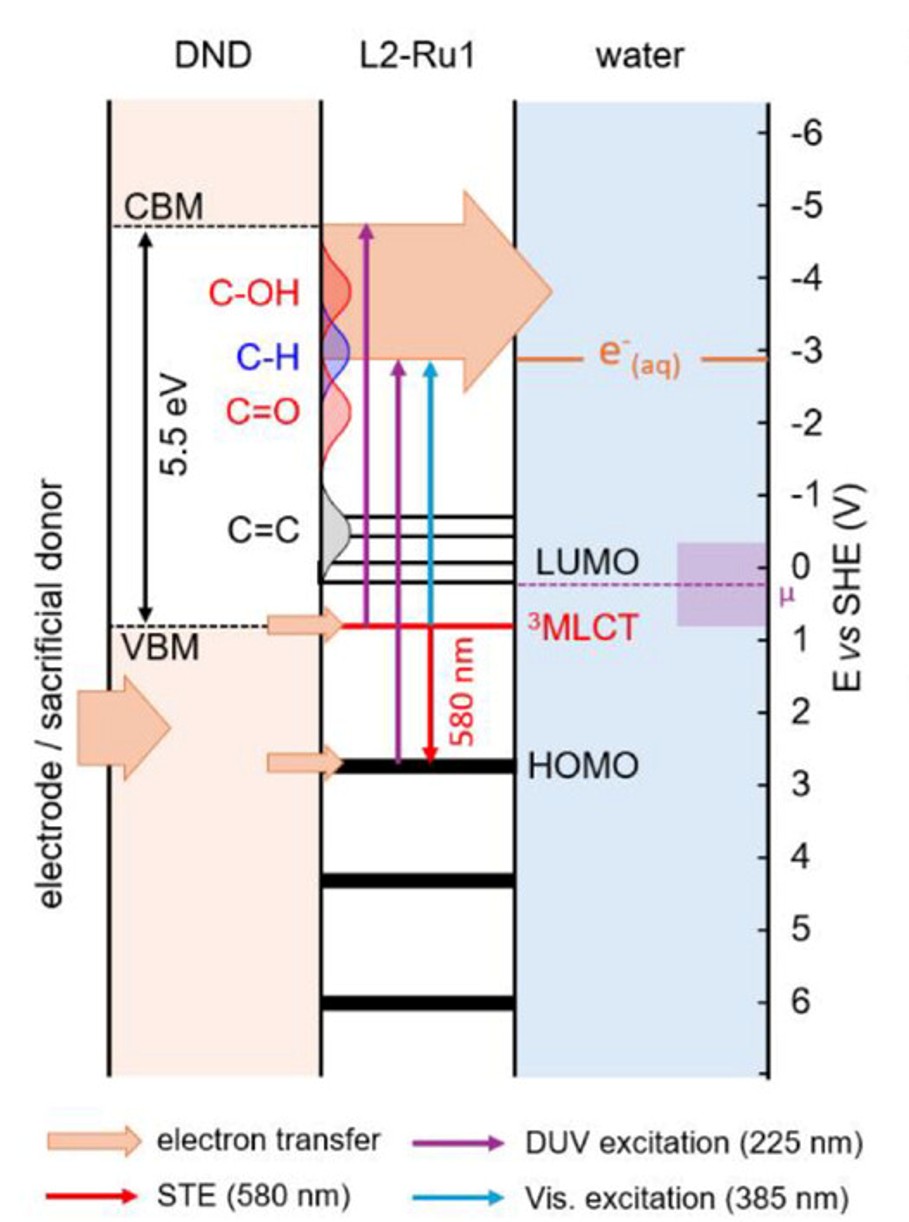
The relevance of this concept is demonstrated by a model photocatalytic reaction: the reduction of CO₂ into value-added molecules for industry. When deposited on electrodes, these functionalized nanodiamonds produce up to 0.76 µmol cm⁻² h⁻¹ of formate under solar illumination in ionic-liquid-based media, nearly twice the activity observed with non-functionalized nanodiamonds. These performances are comparable to, and in some cases exceed, those reported for other diamond-based systems for CO₂ reduction, confirming the potential of this photosensitization strategy.
Beyond this proof of concept, these results open promising perspectives for the development of diamond-based photocatalysts capable of directly harnessing solar energy to drive challenging reduction reactions. Future work will focus on designing more robust photosensitizers and optimizing reaction conditions and reactor architectures to further improve the overall efficiency of these photocatalytic systems.
Reference
« Intrabandgap States Engineering in Functionalized Nanodiamond to Generate Solvated Electrons for Photocatalysis Under Solar Illumination », Advanced Functional Materials, 2025.
Collaboration
- Institut Lumière Matière (UMR5306 Université Claude Bernard Lyon 1, CNRS), Villeurbanne, France.
- Institute of Organic Chemistry, Université de Stuttgart, Allemagne.
- Chemistry Research Laboratory, Université d’Oxford, Royaume-Uni.
- Ångström Laboratory forInorganic Chemistry, Université d’Uppsala, Suède.
- IOLITEC (Ionic Liquids Technologies) GmbH, Heilbronn, Allemagne.
- Center For Integrated Quantum Science and Technology (IQST), Stuttgart, Allemagne.
Contact
- Hugues Girard, Research Director, Laboratoire Édifices Nanométriques – LEDNA, Nanoscience and Innovation for Materials, Biomedicine and Energy – NIMBE, CEA-IRAMIS.
- Jean-Charles Arnault, Research Director, Laboratoire Édifices Nanométriques – LEDNA, Nanoscience and Innovation for Materials, Biomedicine and Energy – NIMBE, CEA-IRAMIS.