

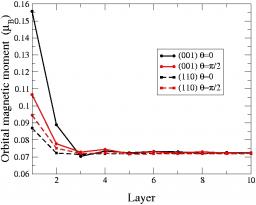
Fig.1 Variation of the component of the local orbital magnetic moment on the magnetization direction as a function of the atomic layer in the (110) and (001) slabs (20 layers) for a magnetization perpendicular (θ=0) or parallel to the surface (θ=π/2).
The magnetic properties (spin and orbital magnetic moments, magneto-crystalline anisotropy energy (MAE)) of nanoparticles, thin films and wires have recently attracted a lot of attention due to their potential applications mainly in the Information Technology sector.
We have carried out a systematic study of the magnetic properties of iron in the tight-binding approximation including s, p, and d valence atomic orbitals taking into account spin polarisation using a Stoner-like model and in the presence of spin-orbit coupling[1]. The validity of the model has first been established by studying in details the magnetic properties of the bcc and fcc bulk phases and comparing the results to those of ab-initio calculations using the PWscf code. This model has then been applied to investigate the (110) and (001) bcc surfaces. The corresponding electronic structures and magnetic moments are very similar to those obtained from ab-initio codes. In addition we have studied the variation of the component of the orbital magnetic moment on the spin quantisation axis as a function of depth, revealing a significant enhancement in the first two layers, especially for the (001) surface (see Fig.1).
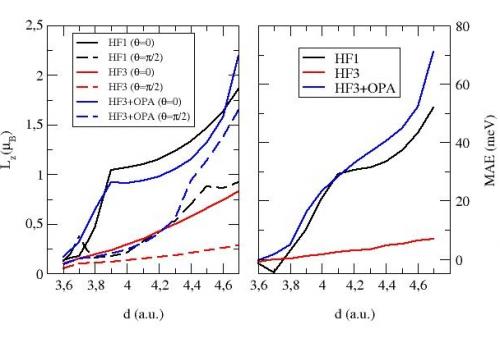
Orbital magnetic moment and magnetocrystalline anisotropy energy for a monatomic Fe wire, at the equilibrium interatomic distance according to three models: full Hartree-Fock (HF1), Stoner-like without (HF3) and with (HF3+OPA) orbital polarization ansatz. θ is the angle between the wire and the magnetization
•  Électronique et optique du futur › Structure électronique et modélisation atomistique
Électronique et optique du futur › Structure électronique et modélisation atomistique
• Laboratory of Physics and Chemistry of Surfaces and Interfaces • UMR 3680 - Service de Physique de l'Etat Condensé (SPEC) • Service de Physique et Chimie des Surfaces et des Interfaces
• Laboratory of Nano-Objects and Complex Systems (LNOSC) • Groupe Modélisation et Théorie / Modeling and Theory Group • GMT-MSIN : Modélisation des Surfaces Interfaces et Nanostructures
