Maëva Almeida, Daniel Dudzinski, Catherine Amiel, Jean-Michel Guigner,Sylvain Prévost, Clémence Le Coeur and Fabrice Cousin, Molecules 28(11) (2023) 4336.
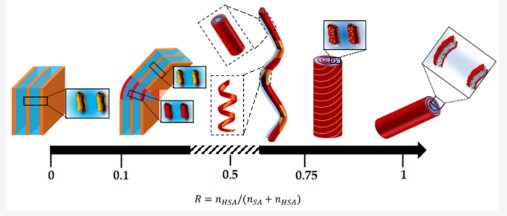
Here, we describe the behavior of mixtures of stearic acid (SA) and its hydroxylated counterpart 12-hydroxystearic acid (12-HSA) in aqueous mixtures at room temperature as a function of the 12-HSA/SA mole ratio R. The morphologies of the self-assembled aggregates are obtained through a multi-structural approach that combines confocal and cryo-TEM microscopies with small-angle neutron scattering (SANS) and wide-angle X-ray scattering (WAXS) measurements, coupled with rheology measurements. Fatty acids are solubilized by an excess of ethanolamine counterions, so that their heads are negatively charged. A clear trend towards partitioning between the two types of fatty acids is observed, presumably driven by the favorable formation of a H-bond network between hydroxyl OH function on the 12th carbon. For all R, the self-assembled structures are locally lamellar, with bilayers composed of crystallized and strongly interdigitated fatty acids. At high R, multilamellar tubes are formed. The doping via a low amount of SA molecules slightly modifies the dimensions of the tubes and decreases the bilayer rigidity. The solutions have a gel-like behavior. At intermediate R, tubes coexist in solution with helical ribbons. At low R, local partitioning also occurs, and the architecture of the self-assemblies associates the two morphologies of the pure fatty acids systems: they are faceted objects with planar domains enriched in SA molecules, capped with curved domains enriched in 12-HSA molecules. The rigidity of the bilayers is strongly increased, as well their storage modulus. The solutions remain, however, viscous fluids in this regime.