Laboratoire Léon Brillouin
UMR12 CEA-CNRS, Bât. 563 CEA Saclay
91191 Gif sur Yvette Cedex, France
+33-169085241 llb-sec@cea.fr

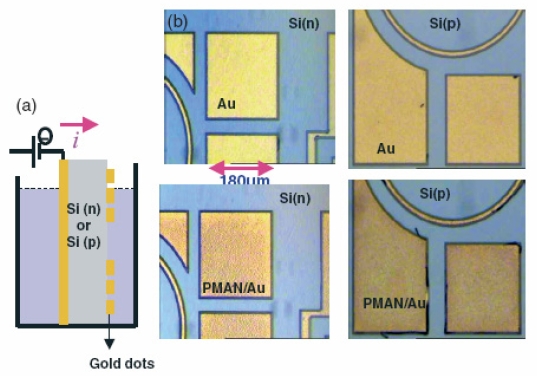
Fig. 1 : (a) Schematic view of patterned gold dots on doped Si substrates; (b) pictures of bare gold dots (top) and grafted gold dots (bottom) after electrografting of pMAN at -3 V vs Ag/Ag+. No grafting was observed by XPS on the silicon areas.
The localized grafting, at a micronic or submicronic scale, of organic substances on surfaces is often a prerequisite in the design of bioelectronics devices, and a valuable component of some combinatorial screening strategies. Moreover, microelectronic devices including transistors, sensors or memories are generally based on interfaces between conducting and quasi-insulating domains resulting from lithographic steps, ionic implantation and silicon oxide thermal growth. Most of these techniques require many steps of transformation and their implementation is often expensive.
We developed a one step technique that allows the localized electrografting of organic matter on designed areas of a composite conducting surface. This method relies on the local work function of electrons, which can easily be tuned by patterning different conducting materials on the substrate. [1-2]
The method was originally demonstrated through examples of composite gold/silicon substrates, which underwent organic electrografting only on gold parts, even when the potential was applied via the silicon substrate and the gold parts were geometrically isolated from the voltage source. That effect is due to:
• The mechanism of cathodic electrografting, which is initiated by an adsorbed radical-anion resulting from a direct electron-transfer from the cathode to electro-active molecules
• The weaker work function of gold with respect to silicon (in solution), which leads to more efficient "output" kinetics from the golden areas.
In a second part of the work, we investigated locally doped silicon surfaces as "composite" substrates to localize cathodic electrografting. We expected to take similar benefit of the higher conductivity of doped areas to promote electron transfer towards the solution and thus initiate the electrografting in a localized manner.
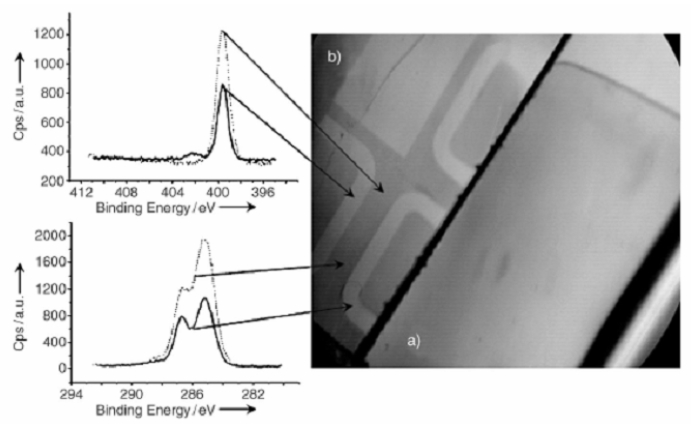
The results were actually the opposite: pMAN electrografted films were thicker on undoped regions of the composite substrates. Moreover, no localization was observed when silicon oxide was removed (by HF treatment) before the electrografting experiment (Fig. 2). Those results were confirmed by XPS, IRRAS, profilometry and TOF-SIMS profiling. [3]
- 90 -
Those surprising results actually arise from the conjugation of two factors :
• The effect of the native silicon dioxide layer: indeed, it is well-known that silicon oxide grows more quickly on highly doped silicon than on poorly doped or intrinsic silicon areas. We can thus consider the silicon oxide layers located on top of highly P-implanted silicon to be slightly more insulating than those located on top of non implanted Si. This would account for the observed results, since an extra-ohmic resistance in the electrical cell should raise the actual cathodic potential experienced by the corresponding area of the substrate. Thus, the implanted silicon areas generate less grafted radical anions than the non implanted ones, which gives rise to significantly thinner grafted films.
• The applied potential (-3.2 V vs. Ag+/Ag), which drives the entire silicon substrate in an accumulation regime and overwhelms the effect of doping when silicon oxide has been removed. That explains why no localization was observed on the "composite" substrates after HF treatment.
•  Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › From molecules to molecular materials
Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › From molecules to molecular materials
• IRAMIS: Saclay Institute of Matter and Radiation • Laboratory of Physics and Chemistry of Surfaces and Interfaces • UMR 3685 NIMBE : Nanosciences et Innovation pour les Matériaux, la Biomédecine et l'Énergie • Service de Physique et Chimie des Surfaces et des Interfaces
• Laboratory of Chemistry of Surfaces and Interfaces (LCSI) • Laboratoire d'Innovation en Chimie des Surfaces et Nanosciences (LICSEN) • Laboratoire Innovation, Chimie des Surfaces Et Nanosciences- LCSI (LICSEN-LCSI)