Laboratoire Léon Brillouin
UMR12 CEA-CNRS, Bât. 563 CEA Saclay
91191 Gif sur Yvette Cedex, France
+33-169085241 llb-sec@cea.fr

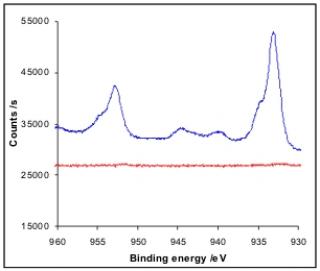
XPS spectra of a PAA-grafted gold substrate accumulated Cu2+ and of the sample after electrolysis (from Ref 2)
Absorption on resins is often used as secondary step in the treatment of water-based effluents, in order to reach very low concentrations. The separation of the trapped effluents from the resins and the regeneration of the resins for further use create wide volumes of secondary effluents coming from the washings of the resins with chemical reagents. We propose an alternative solution based on a « surface strategy » through adsorption phenomena and electrical control of the expulsion stage. The final goal is to limit or ideally to avoid the use of chemical reagents at the expulsion (or regeneration) stage of the depolluting process.
Heavy metal ions were captured on active filters composed by a conducting surface covered by poly-4-vinylpyridine (P4VP) or polyacrylic acid (PAA). Due to pyridine or carboxylic acid groups, those polymer films have chelating properties for copper ions. Our strategy for electrical triggering of the copper expulsion in aqueous medium is based on pH sensitive chelating groups. Applying moderate electro-oxidizing conditions generates acidic conditions in the vicinity of the electrode i.e. “inside” the polymer film. This allows a “switch-off” of the complexing properties of the film from the basic forms (pyridine or acetate) to the acid ones (pyridinium or acid). Interestingly, no buffer washing is necessary to restore (or “switch-on”) the complexing properties of the polymer film because the pH of the external medium is left unchanged by the electrochemical effect that affects only the vicinity of the electrode. Switch-on/switch-off cycles are followed and attested by IR spectroscopy and EQCM method. XPS was also used to confirm the complexation and expulsion steps.
In order to evaluate the concepts detailed in the previous paragraphs, depollution of real aqueous waste was investigated at medium-size volumes (typically a few litres per hour). This project is part of the development of the CENEC process. (CENEC is a French acronym for "Purification process by NanoFiltration and Electro-induced complexation"). CENEC gathers two processes in a row:
1. nanofiltration on ceramic membranes as a first step, which brings the ionic concentration down to ~ 10mg/L. That part of the project is undertaken by LFSM (CEA/DEN/ Pierrelatte);
2. Active filtration on electrically-triggered organic films, which is developed by us to reach ionic concentrations below the mg/L limit. The capture of ionic waste is achieved by spontaneous complexation within grafted organic films. As first examples, non-selective polymers were used, in order to test both the stability of the complexing films upon successive capture-release cycles and the efficiency of the electrically-triggered release. Poly-4-vinylpyridine (P4VP) and polyacrylic acid (PAA) were tested with copper, zinc and nickel solutions at 10 mg/L. In order to reach high capture capacities, carbon felts with specific surfaces higher than 0.6 m2/g were used as conducting substrates. In a second part of this project, a pre-industrial depollution pilot was designed and made in the lab. That pilot will be used as a scale-up exercise, which will allow us to detect and hopefully solve all the technical problems arising from the process itself : draining of gases resulting from the water electrolysis in the vicinity of the carbon felt electrodes, homogeneity of the applied potential on large electrodes, possible electrodeposition on cathodes… The pilot cell has a developed surface of 12 m2. The first tests lead to a capture capacity of 2 liters at 10 mg/L (for a typical waste). The concentration factor obtained with this cell is 8 (The concentration factor is calculated from the diluted “in” and the concentrated “out” volumes). It is noteworthy that the concentration factor improves after each cycle of treatment. The absence of secondary waste allows to take full benefit of this effect.
Contact
References:
(1) Le, X. T.; Viel, P.; Palacin, S.; Electrochemical-pH-switchable polymer film for treatment of metallic ion aqueous waste: from fundamental study to semi-pilot industrial applications; In Electrolysis: Theory, Types and Applications; Kuai, S., Meng, J., Eds.; Nova Sciences Publishers: 2009.
(2) Le, X. T.; Viel, P.; Palacin, S.; Electrochemical behavior of polyacrylic acid coated gold electrodes: an application to remove heavy metal ions from wastewater; Electrochimica Acta 2009, 54, 6089-6093.
(3) Le, X. T.; Viel, P.; Jegou, P.; Palacin, S.; Electrochemical-switchable polymer film: an emerging technique for treatment of metal ion waste water; Separation Purification Tech. 2009, 69, 135-140.
(4) Le, X. T.; Jegou, P.; Viel, P.; Palacin, S.; Electro-switchable surfaces for heavy metal waste treatment: study of polyacrylic acid films grafted on gold surfaces; Electrochem. Comm. 2008, 10, 699-703.
(5) Viel, P.; Dubois, L.; Lyskawa, J.; Sallé, M.; Palacin, S. ; New concept to remove heavy metals from liquid waste based on electrochemical ph-switchable immobilised ligands.; Appl. Surf. Sci. 2007, 253, 3263-3269.
(6) Lyskawa, J.; Sallé, M.; Balandier, J.-Y.; Le Derf, F.; Levillain, E.; Allain, M.; Viel, P.; Palacin, S.; Monitoring the formation of TTF dimers by Na+ complexation; Chem. Commun. 2006, 2233-2235.
(7) Lyskawa, J.; Oçafrain, M.; Trippé, G.; Le Derf, F.; Sallé, M.; Viel, P.; Palacin, S.; Tetrathiafulvalene-based podands bearing one or two thiol functions : Immobilization as self-assembled monolayers or polymer films, and recognition properties.; Tetrahedron 2006, 62, 4419-4425.
(8) Lyskawa, J.; Le Derf, F.; Levillain, E.; Mazari, M.; Sallé, M.; Dubois, L.; Viel, P.; Bureau, C.; Palacin, S.; Univocal Demonstration of the Electrochemically Mediated Binding of Pb2+ by a Modified Surface Incorporating a TTF-Based Redox-Switchable Ligand; J. Am. Chem. Soc. 2004, 126, 12194-12195.
9) Viel, P.; Palacin, S.; Descours, F.; Le Derf, F.; Lyskawa, J.; Sallé, M.; Electropolymerized poly-4-vinylpyridine for removal of copper from wastewater; Appl. Surf. Sci. 2003, 212-213, 792-796.
•  Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › From molecules to molecular materials
Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › From molecules to molecular materials
•  Institut Rayonnement Matière de Saclay • Laboratory of Physics and Chemistry of Surfaces and Interfaces • UMR 3685 NIMBE : Nanosciences et Innovation pour les Matériaux, la Biomédecine et l'Énergie • Service de Physique et Chimie des Surfaces et des Interfaces
Institut Rayonnement Matière de Saclay • Laboratory of Physics and Chemistry of Surfaces and Interfaces • UMR 3685 NIMBE : Nanosciences et Innovation pour les Matériaux, la Biomédecine et l'Énergie • Service de Physique et Chimie des Surfaces et des Interfaces
• Laboratory of Chemistry of Surfaces and Interfaces (LCSI) • Laboratoire d'Innovation en Chimie des Surfaces et Nanosciences (LICSEN) • Laboratoire Innovation, Chimie des Surfaces Et Nanosciences- LCSI (LICSEN-LCSI)




