We study oxide band structure, surface and interface chemistry, ferroelectric and multiferroic films.
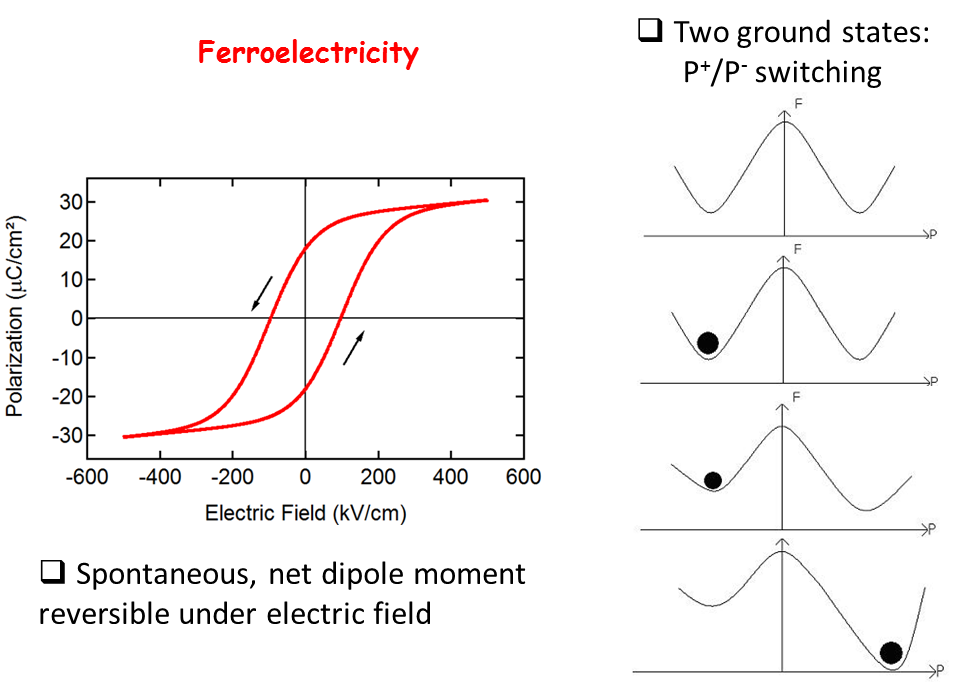
The defining property of a Ferroelectric (FE) material is a spontaneous macroscopic polarization which can be reversed under an applied electric field. The polarization as a function of applied electric field exhibits a hysteresis loop, analogous to ferromagnetic materials, hence the name ferroelectricity.
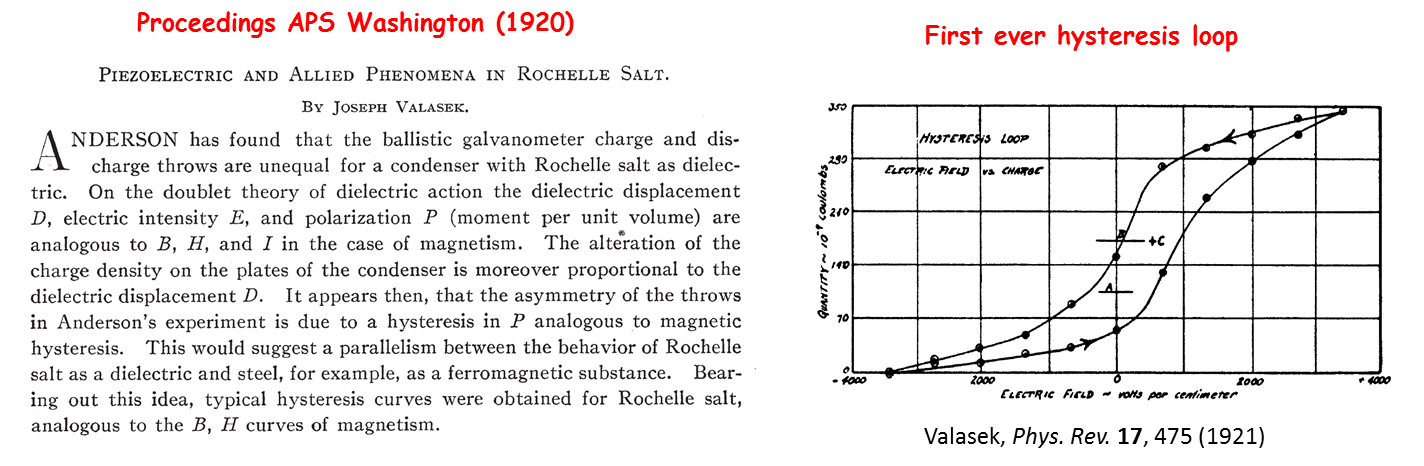
It was discovered by Valasek from the University of Minnesota who presented his results at the Washington meeting of the American Physical Society in April 1920.
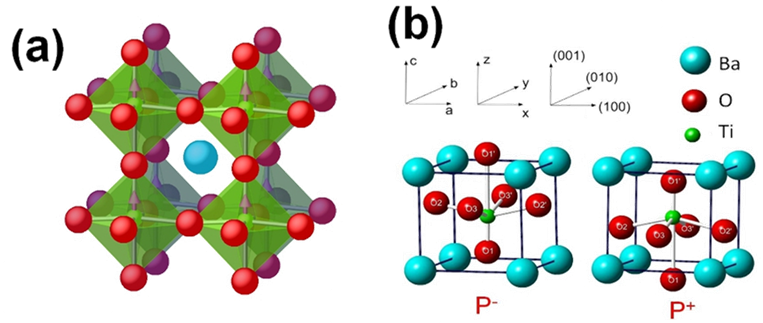
Perovskite oxides, of general formula ABO3 with a pseudocubic structure, where A and B are two different cations, furnish many interesting ferroelectrics. The B-type cation is octahedrally coordinated with oxygen. In the example shown, BaTiO3, it is the relative symmetry breaking displacement of the Ti atoms with respect to the O atoms which is responsible for the spontaneous polarization. BaTiO3 has three ferroelectric phases: tetragonal, orthorhombic and rhombohedral.
•  Les archives de l'IRAMIS et du DRECAM / Archives of DRECAM and IRAMIS › Matériaux, surfaces et nanostructures
Les archives de l'IRAMIS et du DRECAM / Archives of DRECAM and IRAMIS › Matériaux, surfaces et nanostructures  Physique, chimie, nanosciences et matériaux autour des grands instruments
Physique, chimie, nanosciences et matériaux autour des grands instruments
• Laboratory of Physics and Chemistry of Surfaces and Interfaces • Service de Physique et Chimie des Surfaces et des Interfaces
• Laboratory of Nanostructures Studies and Surface Imagery (LENSIS) • Laboratoire d'Etude des NanoStructures et Imagerie de Surface (LENSIS)
