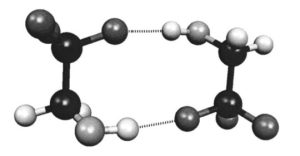
La conformation moléculaire et la liaison H intermoléculaire dans le 2,2,2 trifluoroéthanol (TFE) liquide ont été étudiées par diffraction des neutrons et DFT.
Imre Bakó; Tamás Radnai; Marie Claire Bellisent Funel
The molecular conformation and intermolecular H bonding in liquid 2,2,2 trifluoroethanol (TFE) have been studied by neutron diffraction with hydrogen/deuterium isotopic substitution at room temperature. For comparison, conformations of molecules and their dimers in the gas phase have also been calculated, based on the density functional theory. Energies, geometry, and vibrational frequencies of dimers were analyzed. Diffraction data analyzed by the “Monte Carlo determination of g(r)” (MCGR) method resulted in a molecular structure in agreement with the findings from gas phase electron diffraction experiments and density functional calculations. The intermolecular structure functions were compared to the same functions obtained from a molecular dynamics simulation. All of the composite radial distribution functions are in good agreement with the simulation results. According to our calculation the hydrogen-bonded aggregation size is smaller in pure liquid TFE than in pure liquid ethanol.
Investigation of structure of liquid 2,2,2 trifluoroethanol: Neutron diffraction, molecular dynamics, and ab initio quantum chemical study,
Imre Bakó, Tamás Radnai, Marie Claire Bellisent Funel, J. Chem. Phys. 121 (2004) 12472–12480.