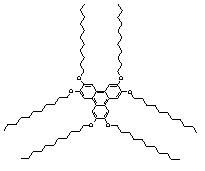
Among the many types of liquid crystals (LCs), planar disk-like molecules such as triphenylenes are known to exhibit columnar mesophases. An example of such compounds are 2,3,6,7,10,11-hexakis-alkoxy-substituted triphenylenes, hereafter noted Tn where n indicates the number of carbon atoms in the alkyl chains. Tn molecules consist in flat polyaromatic cores symmetrically surrounded by flexible n-carbon side chains (Figure 1). When deposited on a surface, Tn molecules stack upon each other to form hexagonally packed columns with a p-p stacking configuration. Triphenylene LC mesophases present a quasi-1D transport of charge carriers along these vertical columns which make them attractive for electronic devices applications.
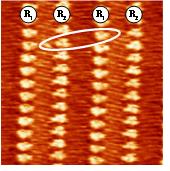
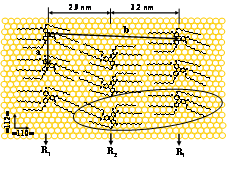
In order to control the 3D arrangement of Tn molecules on a metal surface, it is necessary to control the structure of the first Tn monolayer. We report here on in-situ STM investigations of T11 (Figure 2) self-assembled monolayers (SAMs) at the n-tetradecane/Au(111) interface with the essential aim to elucidate the interplay between substrate-molecule and molecule-molecule interactions.
Shortening the length of the six peripheral alkyl chains strongly affects the 2D arrangement of Tn on gold. For chains as short as T5 and T7, molecules adopt a hexagonal packing instead of twin rows. Surprisingly, for an intermediate chain length such as T9, both types of packing coexist in the same monolayer, a clear case of symmetry breaking which we attribute to a lever arm effect of the chains (see Ref.2). In conclusion, tuning the length of the alkyl chain appears as an efficient way to precisely control the 3D arrangement of triphenylenes Tn in columnar discotic on conducting surfaces.
REFERENCES:
1. N. Katsonis, A. Marchenko, D. Fichou, J. Am. Chem. Soc. 2003.
2. N. Katsonis, A. Marchenko, D. Fichou, Eur. Phys. J. B submitted.
•  Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › From molecules to molecular materials
Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › From molecules to molecular materials  Future optics and electronics › Organic and molecular electronics
Future optics and electronics › Organic and molecular electronics
• Service de Physique et Chimie des Surfaces et des Interfaces