
Functionalization of Carbon Nanotubes through Polymerization in Micelles
Our method is based on the controlled polymerization of hydrophobic molecules on nanotubes dispersed in micelles. This approach permits to obtain carbon nanotube hybrids exhibiting high stability while preserving their π-conjugated system responsible for their outstanding optical and electrical properties. The nanotube hybrids can be purified, manipulated and dispersed in various solvents without loss of their functionality. Extensive characterizations based on optical and microscopic measurements demonstrate the strength of this method for designing new functional materials.
During the last decade, the rapid rise of nanosciences brought new paradigms for developing renewable energy sources and lowering the consumption of electronic devices. For many applications, the fabrication of hybrid materials (e.g. organic/inorganic, organic/nano, nano/bio, etc...) is required. Thus the control over the association of two chemically different subunits has to be performed with methods that can preserve the integrity and functionality of the building blocks. Thanks to their outstanding physical and chemical properties, carbon-based nanomaterials (carbon nanotubes, graphene) are among the most promising materials for future technologies.
It is well established that the covalent grafting of molecules onto the nanotube sidewalls give rise to robust conjugates since the nanotubes and the addends are linked through covalent bonds; however, the transformation of carbon atoms hybridized sp2 into sp3 in the nanotube framework induces a sizeable loss of their electronic properties. On the contrary, the non-covalent functionalization permits to better preserve the electronic properties of the nanotubes. So, for a number of applications, the non-covalent functionalization should be preferred. However, this approach suffers from a major drawback which is the lack of stability of the resulting assemblies. Indeed, molecules adsorbed onto the nanotube sidewall can desorb, more or less easily, when for example the solvent changes or the nanotubes are filtered and redispersed. Therefore there is a real needed of methods combining the advantages of the covalent and non-covalent methods without their respective drawbacks.
Recently, we described an innovative method of functionalization of carbon nanotubes and graphene combining most advantages of the covalent and non-covalent approaches without their principal drawbacks. Our method is based on the non-covalent association of functional monomeric molecules on carbon nanotubes or graphene dispersed in micelles followed by the polymerization of the monomeric molecules inside the micelles to give rise to core-shell structures exhibiting high chemical stability and good optical properties.(1)
- Clavé, G.; Delport, G.; Roquelet, C.; Lauret, J.-S.; Deleporte, E.; Vialla, F.; Langlois, B.; Parret, R.; Voisin, C.; Roussignol, P.; Jousselme, B.; Gloter, A.; Stephan, O.; Filoramo, A.; Derycke, V.; Campidelli, S. Chem. Mater. 2013, 25, 2700-2707.
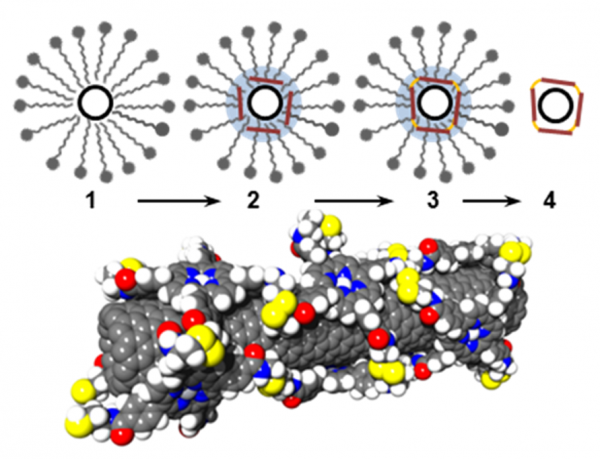
Schematic representation of the different steps of functionalization: 1) the nanotubes are dispersed and individualized in micelles; 2) porphyrins in an organic solvent are introduced on the nanotube surfaces; 3) after solvent evaporation, the reaction to cross-link the porphyrin is performed leading to a stable organic shell around the nanotubes; 4) the nanotube derivatives are purified via filtration and extensive washing to remove the surfactants, reagents and unbound porphryins.
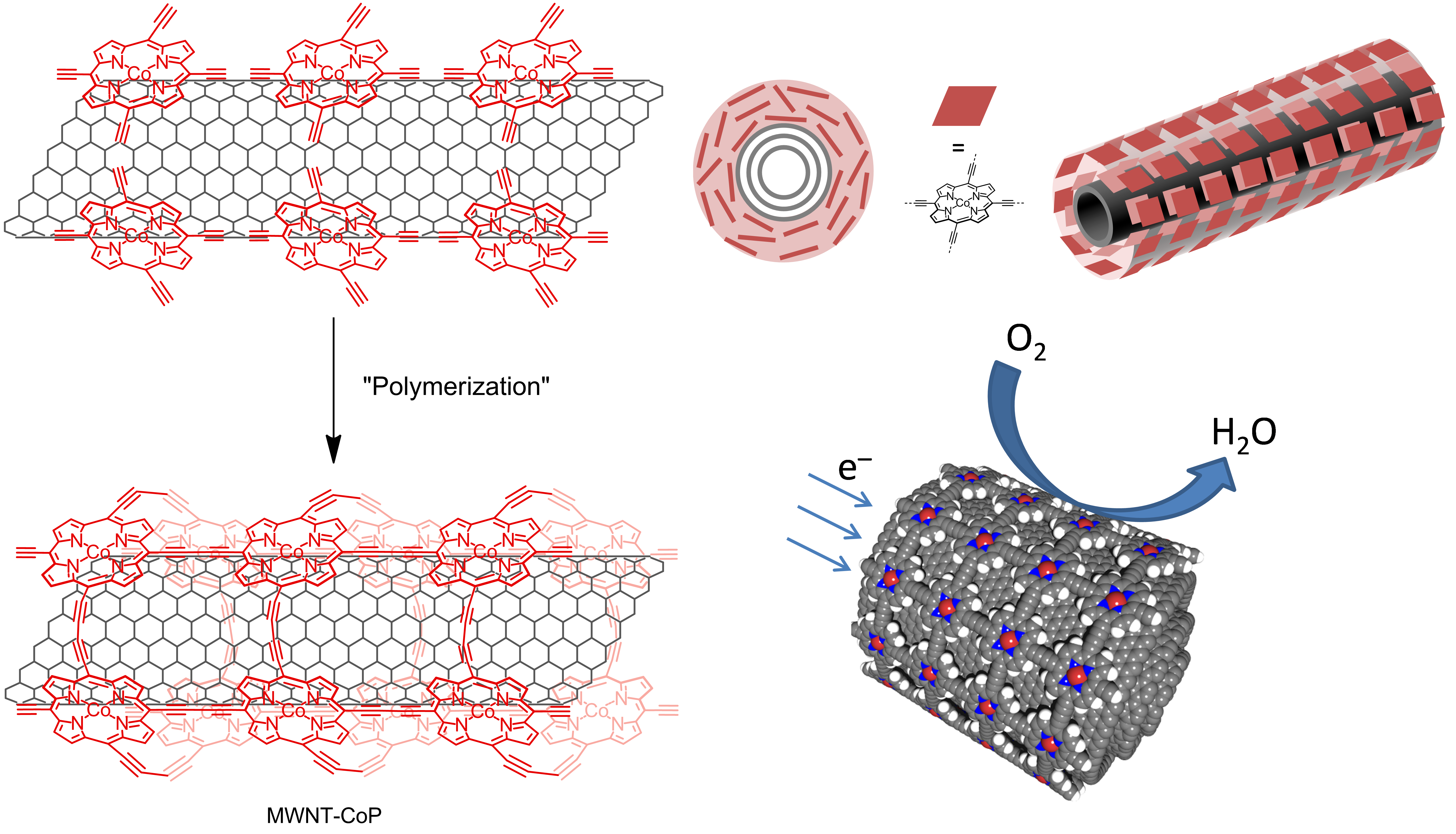
Schematic representation of templated polymerization around the nanotubes and their use for oxygen reduction reaction
Carbon Nanotube-Templated Synthesis of Covalent Porphyrin Network for Oxygen Reduction Reaction
Within the context of sustainable development and renewable energy, we and others envisioned the use of functionalized or doped carbon nanotubes in electrocatalytic systems. In such systems, the catalytic sites, which can be made of metallic nanoparticles, organometallic complexes or any chemical functional groups, need to be supported on conducting materials. Carbon nanotubes, thanks to their electrical conductivity and their high surface area appear as the ideal material for that purpose.
We developed a method of functionalization of carbon nanotubes based on the adsorption of cobalt(II) meso-tetraethynylporphyrins on the nanotube sidewalls followed by the dimerization of the triple bonds via Hay-coupling;(1) during the reaction, the nanotube acts as a template for the formation of the polymeric layer. The material shows an increased stability resulting from the cooperative effect of the multiple π-stacking interactions between the porphyrins and the nanotube and by the covalent links between the porphyrins. The nanotube/porphyrin hybrids were tested as the supported catalyst for the oxygen reduction reaction (ORR) in a series of electrochemical measurements under acidic conditions. Compared to similar systems in which monomeric porphyrins are simply physisorbed, MWNT−CoP hybrids showed a higher ORR activity associated with a number of exchanged electrons close to four, corresponding to the complete reduction of oxygen into water.(2)
- Hay, A. S. J. Org. Chem. 1962, 27, 3320-3321.
- Hijazi, I.; Bourgeteau, T.; Cornut, R.; Morozan, A.; Filoramo, A.; Leroy, J.; Derycke, V.; Jousselme, B.; Campidelli, S. J. Am. Chem. Soc. 2014, 136, 6348-6354.
•  Électronique et optique du futur › Matériaux pour l'électronique et l'optique - Electronique organique et moléculaire
Électronique et optique du futur › Matériaux pour l'électronique et l'optique - Electronique organique et moléculaire
• Laboratoire d'Innovation en Chimie des Surfaces et Nanosciences (LICSEN)





