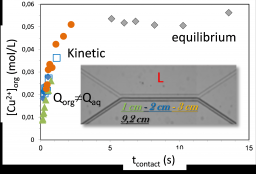
Kinetic extraction of copper salt by HDEHP/decanol solution for a PDMS-glass Y-shape geometry. The time dependence is obtained by general flow variation with constant Qorg/Qaq=10 ratio.
In the framework of an internal CEA program dedicated to the liquid-liquid extraction for nuclear waste treatment (or primary extraction of uranyl), we have fully characterized the structure of the organic phase with its influence on the phase stability[1],[2] and more recently on the extraction efficiency[3]. Extraction is related to solubilization properties[4] where surfactant and hydrotrope can be used in closed connection to enhance the transfer[5]. In such processes, the liquid-liquid interface plays a key role even though its specific influence is not fully understood. Recently, we took benefit of microfluidic tools to control the interface by making the interfacial reactions predominating over bulk ones. Dedicated home-made chips have shown the possibility to follow kinetic but also to control the shearing properties of the interface
[1] Chapter 7 for Ion Exchange and Solvent Extraction F. Testard, P. Bauduin, L. Berthon, Th. Zemb. Ed B. Moyer, CRC Press, chap 7, P381 (2009).
[2] Berthon L, Testard F, Martinet L, Zemb T, Madic C., C. R. Chimie (2010), 13 1326–1334
[3] Dourdain, S; Hofmeister, I; Pecheur, O; Dufreche, JF; Turgis, R; Leydier, A; Jestin, J; Testard, F; Pellet-Rostaing, S; Zemb, T Langmuir (2012), 28 11319-11328.
[4] Kunz, W., Testard, F., Zemb, Th., Langmuir, (2009), 25(1), 112-115.
[5] Bauduin, P., Zemb, Th., Testard, F., J. Phys. Chem. B (2008) 112, 12354-12360.
An alternative to classical liquid-liquid extraction is the cloud point extraction where a condensed phase separate spontaneously under a stimuli. In collaboration with yyChantal Larpent from Institut Lavoisier (UVSQ), new thermo-responsive metal-chelating surfactants were developed and used for uranyl cloud point extraction[1],[2]. These results hold most promise for the development of new solvent-free separation process but also for specific reactivity[3].
[1] Prévost S., Coulombeau H., Baczko K., Zorz N., Desvaux H., Testard F., Zemb Th. And Larpent Ch., Tenside Surf. Det. (2009) 46, 100-104.
[2] Nave, S., Testard, F., Coulombeau, H., Baczko, K., Larpent, C., Zemb, Th., Phys. Chem. Chem. Phys., (2009), 11, 2700-2707.
[3] Thèse Richard Gadou, UVSQ, (2010).
• › Interfaces, fluides complexes et microfluidique  Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects Synthesis and characterization of nano-objects
Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects Synthesis and characterization of nano-objects
• UMR 3299 - Service Interdisciplinaire sur les Systèmes Moléculaires et les Matériaux • Service Interdisciplinaire sur les Systèmes Moléculaires et les Matériaux
• Laboratoire Interdisciplinaire sur l'Organisation Nanométrique et Supramoléculaire (LIONS) • Interdisciplinary Laboratory on Nanoscale and Supramolecular Organization





