The nucleation and growth of three-dimensional superlattices of gold nanoparticles has been followed directly in situ by means of small angle x-ray scattering. These assemblies spontaneously form in a dilute solution providing the particles are large enough to generate a van der Waals driven attraction sufficient to counterbalance the thermal energy. The superlattices nucleate very soon after the birth of the individual particles and their growth kinetics is slower than predicted by a mechanism of simple diffusion of the nanoparticles towards the superlattices. The superlattices are first limited in size (170 nm in diameter) and have a globular shape with a low polydispersity. They present a fcc inner structure with nanoparticles being separated by a capping agent bilayer yielding a low gold internal volume fraction (ϕSL=0.33). In a second stage, these superlattices coalesce with time.
Effect of adhesion/capillary force balance on the aggregation state of adsorbed nanoparticle after drying.
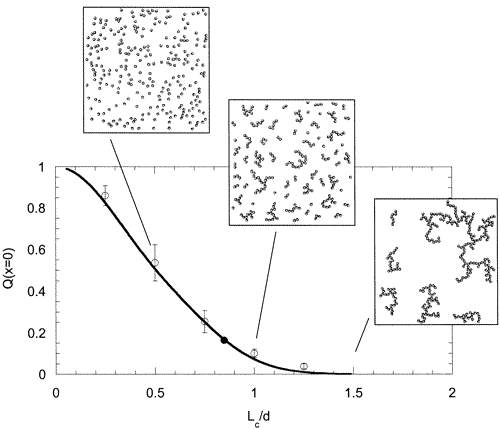
This work deals with the common atomic force microscopy observation that small particles weakly adhering to a substrate tend to form aggregates at least when imaged under air. We show that it is possible to measure the coverage of mica surfaces with α-Al2O3 colloids in situ by means of static light scattering. The dynamic properties of the speckled pattern obtained from the layer are also used to probe the dynamic of the particles. It is then concluded that even the weakest adhesion of the particles does not allow in situ aggregation and surface mobility (at least, on the time scale of the laboratory experiment). The adhering particles aggregate only during the drying stage, due to the action of lateral capillary forces. A model for the aggregation mechanism is introduced taking into account the balance of forces acting on the particles during the drying stage.
When colloids deposited on a macroscopic substrate are imaged after drying, two-dimensional aggregates are commonly observed. When the colloids are initially stable and well dispersed in solution, the final presence (or not) of these surface aggregates is linked to the balance between the mutual colloids–surface interactions and the capillary forces. In this work, we control the adhesion of α-Al2O3 colloids onto mica using a surface molecular ligand (citric acid) and measure the associated structure of the layer both in situ and after drying. The dynamic property of speckle pattern obtained from light scattering of the layer is used to probe in situ the dynamics of the layer. It is therefore concluded that even the weakest adhesion of the particles does no allow in situ surface mobility nor aggregation. The adhering particles aggregate only during the drying stage, due to the influence of lateral capillary forces. A model for the aggregation mechanism is introduced taking into account the balance of forces acting on the particles during the drying stage.
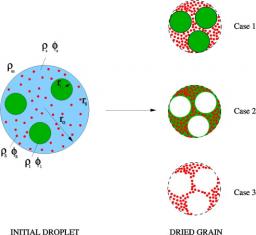
The schematic geometries and scattering length density considered in the grain model. Case 1: intact polybromostyrene particles inside silica particles; case 2: holes inside silica particle and melted polymer; case 3: holes inside silica particles when the polymer is fully calcined.
Nanoporous powders are prepared in a single step by spray-drying mixtures of small inorganic and larger organic nanoparticles. The structure of these powders has been studied as a function of the mixture's composition using silica and polybromostyrene nanoparticles. Scanning electron microscopy reveals the presence of an increasing concentration of mesopores as the concentration of polybromostyrene increases. By coupling thermogravimetric analysis and ultra-small-angle X-ray scattering, the structure is quantitatively linked to the composition of the porous grains. Experimental USAXS intensities are compared to scattering models for the composite powders. It allows to demonstrate that (i) all mesopores are empty even in the center of the grains, (ii) part of the polymer remains in the micropores of the dried grains. A quantification of the fraction of micropores filled by residual polymer is presented. Such a synthesis procedure can be used to produce hydrophobic porous powders in a one-step process.
Nanoparticles confined in droplets of less than a picoliter are forced to organize in submicronic dry grains through solvent evaporation. The evolution of structures of the grains and the constituent nanoparticles during the slow drying process are investigated in situ with small-angle X-ray scattering (SAXS) for the first time. The scattering results have been explained on the basis of the equilibrium thermodynamics of the droplets in the drying tube. We demonstrate that this technique is really efficient in describing the internal arrangement of the nanoparticles inside the drying droplets. Distinction between an almost homogeneous repartition of the nanoparticles in droplets and formation of core shell like particles even in strongly polydispersed droplets can be made using SAXS.
•  Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › Matériaux nanocomposites nanostructurés (cristallisés et matière molle) : de leur élaboration, à leurs propriétés. Synthesis and characterization of nano-objects › Molecular assembly and nanostructured materials
Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects › Matériaux nanocomposites nanostructurés (cristallisés et matière molle) : de leur élaboration, à leurs propriétés. Synthesis and characterization of nano-objects › Molecular assembly and nanostructured materials
• UMR 3299 - Service Interdisciplinaire sur les Systèmes Moléculaires et les Matériaux
• Laboratoire Interdisciplinaire sur l'Organisation Nanométrique et Supramoléculaire (LIONS) • Interdisciplinary Laboratory on Nanoscale and Supramolecular Organization
