Nanoparticles, considered as tools or analytes, have been the focus of much attention in Analitycal Sciences Research these last years. As tools, they are extractants, surface modifiers, etc…. as analytes, they require quantification just as bulk solid or dissolved materials but also physical and chemical characterizations (size, surface properties, shape, …). Our activities cover several aspects:
Quantitative Determination of Total TiO2 in River Water by ICPMS
The LIONS laboratory proposes a new sample preparation method to reliably determine by ICPMS total titanium oxide (particles<320µm, nanoparticles and salts) in the environment. This procedure is applied to real samples (Seine river water). The results obtained are in agreement (better than 3%) with the reference procedure (complete dissolution of insoluble faction). Elements of this sample treatment procedure are the object of a patent.

Patent:
"Procédé de dosage d'un élément présent tout ou partie sous la forme de particules en suspension dans un liquide". French patent n°12 60918 du 16 novembre 2012
Publication: to be submitted
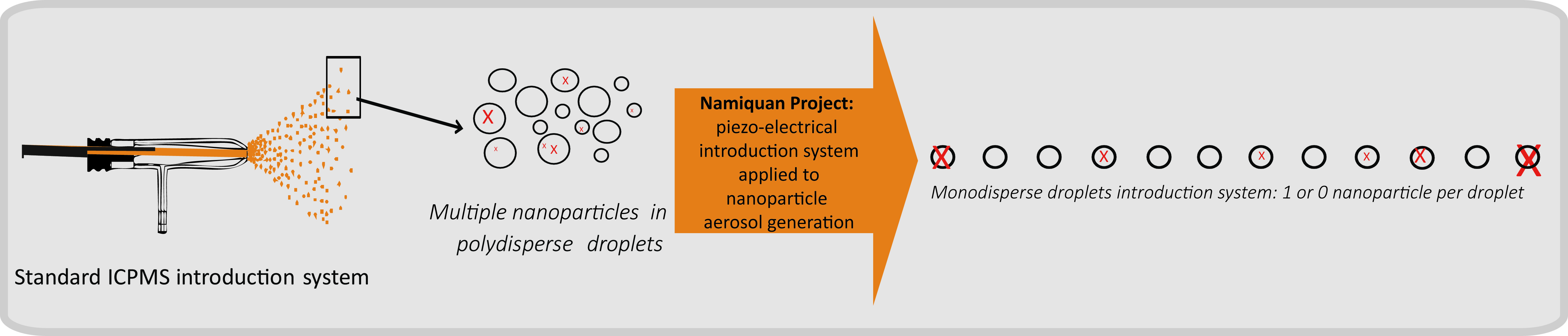
Namiquan Project for ICPMS
This project consists in developing an experimental instrument to characterize nanoparticles suspension in size and elemental composition at low concentration level. It is based on the replacement of a standard ICPMS introduction system (pneumatic nebulizer) by a monodisperse droplets product system. Each droplet is then individually analyzed, as a unique sample. The methodology will first be applied to length and composition polydispersity evaluation of imogolites (Si/Al/Ge ratio). This project is supported by The Région Ile de France DIM Analytics and le triangle de la physique RTRA.
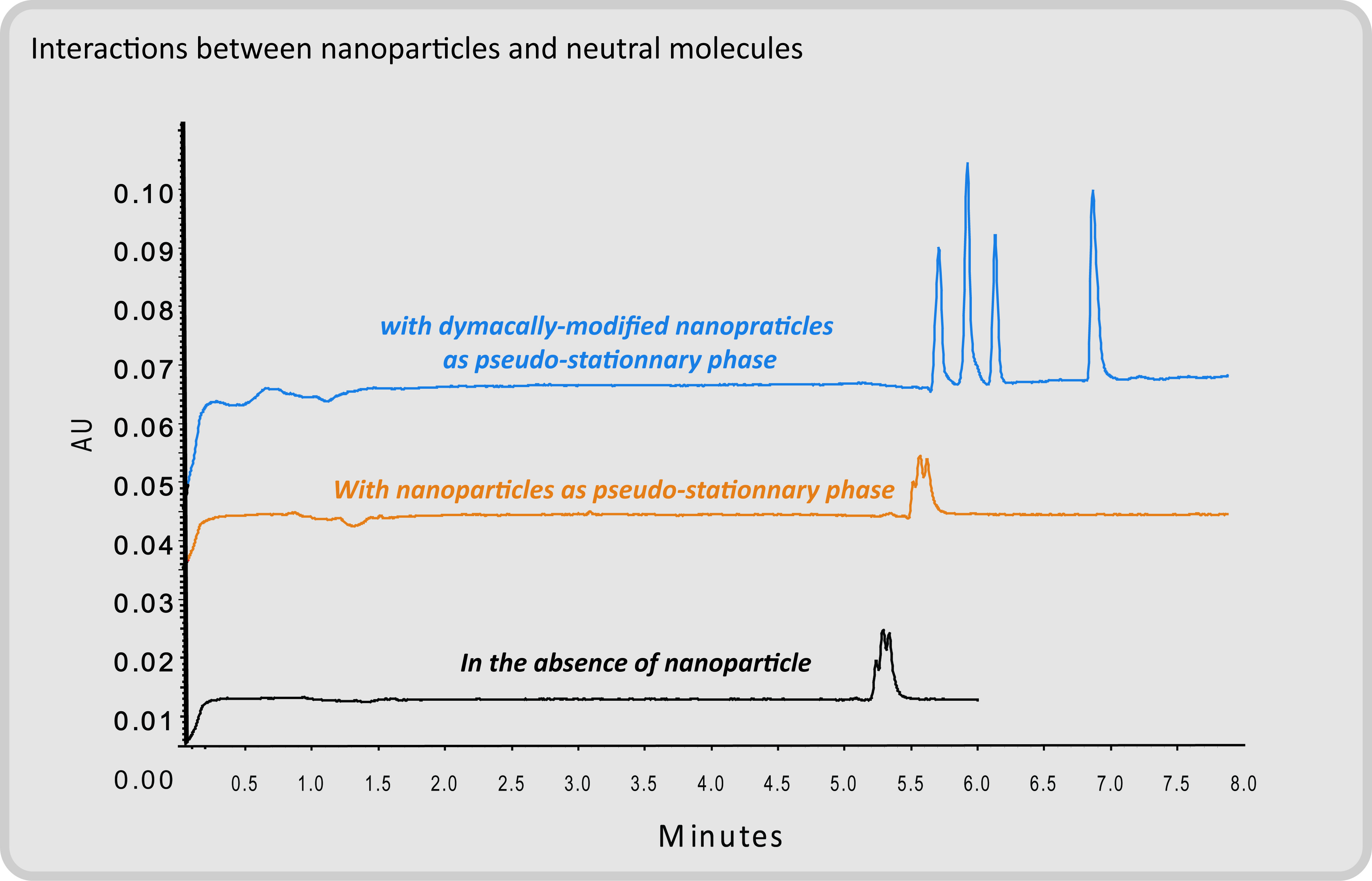
Interactions between Nanoparticles and Molecules by Capillary Electrophoresis
Capillary electrophoresis is a miniaturized separation technique widely used for the separation of charged species according to their mass/charge ratio, whereas neutral compounds migrate with the electroosmotic flow. The introduction of nanoparticles in the electrolyte perturbs both charged and/or neutral species providing access to interactions between these nanoparticles and the solubilized compounds.
• › Nano-chimie, nano-objets / Nano-chemistry, nano-objects  Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects
Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects
• UMR 3299 - Service Interdisciplinaire sur les Systèmes Moléculaires et les Matériaux
• Laboratoire Interdisciplinaire sur l'Organisation Nanométrique et Supramoléculaire (LIONS)
