•  Interaction laser-matière › Physico-Chimie et Chimie-Physique
Interaction laser-matière › Physico-Chimie et Chimie-Physique
• Laboratoire Interactions, Dynamiques et Lasers (LIDYL) - CEA-CNRS et Université Paris Saclay
• LUMO-DyR - Reaction Dynamics Team • LUMO - Equipe Dynamique Réactionnelle (DyR)


Photoionization of Benzophenone in the Gas Phase: Theory and Experiment
N. Khemiri, S. Messaoudi, M. Abderrabba, G. Spighi, M. A. Gaveau, M. Briant, B. Soep, J. M. Mestdagh, M. Hochlaf and L. Poisson
J. Phys. Chem. A, 119, 6148, 2015 [doi]
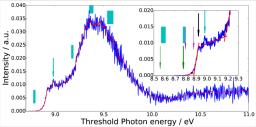
We report on the single photoionization of jet-cooled benzophenone using a tunable source of VUV synchrotron radiation coupled with a photoion/photoelectron coincidence acquisition device. The assignment and the interpretation of the spectra are based on a characterization by ab initio and density functional theory calculations of the geometry and of the electronic states of the cation. The absence of structures in the slow photoelectron spectrum is explained by a congestion of the spectrum due to the dense vibrational progressions of the very low frequency torsional mode in the cation either in pure form or in combination bands. Also a high density of electronic states has been found in the cation. Presently, we estimate the experimental adiabatic and vertical ionization energy of benzophenone at 8.80 +/- 0.01 and 8.878 +/- 0.005 eV, respectively. The ionization energy as well as the energies of the excited states are compared to the calculated ones.
Energetics and ionization dynamics of two diarylketone molecules: benzophenone and fluorenone
Z. Gouid, A. Röder, B. K. Cunha de Miranda, M.-A. Gaveau, M. Briant, B. Soep, J.-M. Mestdagh, M. Hochlaf and L. Poisson
Phys. Chem. Chem. Phys., 21, 14453, 2019 [doi]
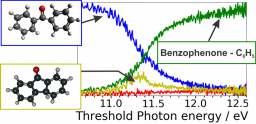
Single photon ionization and subsequent unimolecular ion decomposition were studied on jet-cooled benzophenone and fluorenone separately, using VUV synchrotron radiation in a photoion/photoelectron coincidence setup. Slow PhotoElectron Spectra (SPES) were recorded in coincidence with either the parent or the fragment ions for hν < 12.5 eV. Dissociative ionization is observed for benzophenone only. The full interpretation of the measurements, including the identification of the neutral and ionic species when dissociative ionization is at play, benefits from high level ab initio computations for determining the equilibrium structures and the energetics of the neutral and ionized molecules and of their fragments. Electronically excited states of the parent molecular ions were calculated also. From this analysis, an accurate experimental determination of the energetics of the benzophenone and fluorenone ions and of their fragmentation channels is available: adiabatic ionization energies of benzophenone at 8.923 ± 0.005 eV and of fluorenone at 8.356 ± 0.007 eV; and appearance energies of benzophenone fragment ions at 11.04 ± 0.02 eV (loss of C6H5), 11.28 ± 0.02 eV (loss of H) and 11.45 ± 0.02 eV (loss of CO). The corresponding fragmentation mechanisms are explored, showing likely concerted bonds rearrangement. Possible pre-ionizing fragmentation is discussed in light of the spectra presented. The structural rigidity of fluorenone diarylketone seems to be the origin of the inhibition of the fragmentation of its cation.