

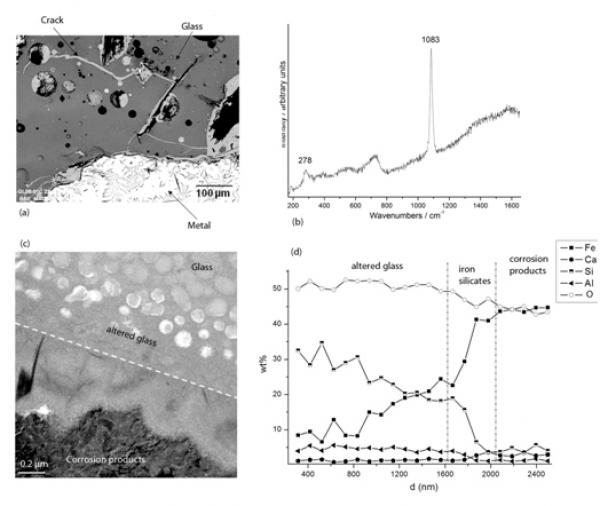
(a) SEM. Ironmaking slag sample containing metal, glass and the crack network filled with corrosion products (b) µRaman spectra of siderite (FeCO3) from the corrosion products, (c) TEM photograph of the glass/corrosion product interface, (d) EDS-STEM chemical distribution (Kα lines) perpendicularly to the interphases.
To gain insight in the long-term behavior of nuclear waste packages at the glass–metal–clay interfaces in their geological repository conditions, slags from ancient ironmaking sites can be used as valuable proxies.
The question of the interaction between glass and iron is particularly important because nuclear glasses are poured in a stainless steel canister and the resulting package is expected to be surrounded by a thick carbon steel overpack (Andra, 2005). Some laboratory experiments under anoxic conditions have shown that the presence of iron and more generally iron corrosion products sustain glass alteration at a rate higher than the glass would undergo in the presence of site water only. This enhancement of glass alteration is due to the strong affinity of iron rich phases with respect to silicon, the main component of the glass (De Combarieu, et al., 2010 in press, Jollivet, et al., 2000). It has been asserted that the formation of Fe-Si containing phase delay the formation of the protective gel layer responsible for the drop of alteration rate observed in glasses altered by water under static or low flow rate conditions (Combarieu, et al., 2010 in press). That is why understanding and modeling interactions between glass, iron and clay is a key preliminary step in order to predict long-term behavior of nuclear glass packages. Consequently, studies of natural or archaeological analogues give precious observational insight for this issue. For example, the site of Glinet dated from the 16th century (Neff, et al., 2010), has been taken as a reference site for studying glass/iron interactions. Indeed, the by-product slags produced by the blast furnace present partly glassy matrix and contain metallic particles of micrometric to millimetric sizes. The blocks present numerous cracks that were immediately saturated with water as they were buried at depths continuously under the water level. It has been verified by on-field measurements and laboratory tests that at the sample location, water was carbonated and anoxic (Saheb, et al., in press). Moreover the crack network connects the metallic zones of the slags to the external solution of the soil and is entirely filled by corrosion products of iron such as those found in desaerated media (Fig. a). Microstructural characterization of the network embedded with the corrosion products by µRaman spectroscopy, reveal the presence of siderite (FeCO3- Fig b). This siderite contains several % of calcium partially substituting the Fe atoms. This substitution was also observed on iron artefacts corroded in the same medium and could come from the groundwater but also, in the present case, from the glass matrix, that contain this element. Actually, the glasses identified in Glinet are calcium silicate glass rich in iron and aluminum (%masMgO: 0.7-2.2, %masAl2O3: 5.1-8.8, %masSiO2: 61.9-76.5, K2O: 1.8-2.3, %masCaO: 15.6-25.1, %masMnO: 0.2-2.9, %masFeO: 1.2-9.1). This range of compositions for the ternary CaO-A2O3-SiO2 defines an area known for its immiscibility (Neuville 2006). SEM-FEG observations confirm a non-homogeneous nanostructure characterized by the presence of spheroids of several hundreds of nanometers distributed in a glass matrix. This specific structure of the glass implies different alteration kinetics for each of the phases observed by TEM (Fig c). The spheroids, mainly constituted of SiO2, are less altered than the embedding matrix containing higher concentration the other elements.
At the interface between glass and iron corrosion products, different layers have been observed, linked to the variation of composition (Figc and Figd). EDS analyses coupled to STEM indicate that the altered glass is depleted in Ca but not in Al. Interestingly, an external festooned layer between altered glass and siderite, made of amorphous iron silicates is observed (Figc). The study of this layer can give insight into the influence of iron on the glass corrosion. Moreover, iron coming from the metal corrosion products has penetrated into the altered layers of the glass. Laboratory experiments simulating a crack, have been conducted for 120 days at 50°C on synthetised glass samples with the structure and the chemical composition of the slag glass matrix. The chemical profiles of the glass alteration layers are very comparable to the archaeological ones and are currently under study, The results are expected to help discriminating between the controlling parameters of glass alteration: transport vs chemical equilibria.
Libourel, G., Verney-Carron, A., Andreasmorlok, Gin, S., Sterpenich, J., Michelin, A., Neff, D., Dillmann, P., 2011. The use of natural and archeological analogues for understanding the long-term behavior of nuclear glasses, Comptes Rendus de l'Académie des Sciences, Géoscience, in press
•  Matériaux des nouvelles technologies pour l’énergie › Corrosion long terme de matériaux métalliques / Long term corrosion of multimaterials containing metals
Matériaux des nouvelles technologies pour l’énergie › Corrosion long terme de matériaux métalliques / Long term corrosion of multimaterials containing metals  Physique, chimie, nanosciences et matériaux autour des grands instruments
Physique, chimie, nanosciences et matériaux autour des grands instruments
• Laboratoire archéomatériaux et prévision de l'altération (LAPA)





