Since the development of the ICP-MS in the years 1980, some applications remain difficult. The problems involved in the determination of the traces in charged matrix persist and the assumption of an intrinsic limitation to the technique was mentioned. Two key parameters of the ICP-MS performances are tolerance with strong contents easily ionisable salts on one hand and refractory oxide formation of the other hand.
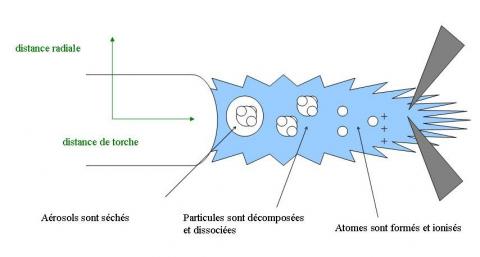
A simplified diagram of the course of an aerosol drop of sample through plasma is represented figure 1. The aerosol crosses plasma very quickly (a few milliseconds). In this time, plasma must dry, break up, dissociate, atomize and ionize the sample with 100% of conversion if possible into monocharged ions. The matrix effect affects the efficiency of this process [1-4] by modifying displacement of ion - atom equilibrium for an analyte, the energy demand, the ion - electron recombinations, the side diffusion, the ambipolar diffusion, the increase in the collisions, the effect of space load.
To correct the matrix effects of the easily ionisable elements (EIE), one can try to recreate the matrix with synthetic solutions and to use it for calibration. However, most often, one does not know exactly the sample composition and this method is not usable any more. It is also possible to make an analysis by proportioned additions but the implementation claims much time [5]. To mitigate the effects of the EIE, certain spectroscopists manufacture ionic plugs, but this method is limited by the extinction due to the effect of load [4]. From our point of view, the best way of correcting these effects is to understand the origin of the modification of the process of ionization in order to adjust the operating conditions to reduce or eliminate the interference caused by the matrix.
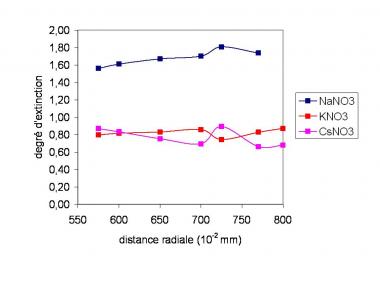
Figure 2: Extinction degree of Al for a distance from torch of 0 with the elements of matrix Na, K and Cs
The most studied matrices are Na, K and Ca not only for historical reasons, since they were largely known in the flame techniques, but also because they are most frequent in the environment (sea waters and river, biological fluids) [1]. To be able to bring closer our results those of the literature and to validate our methodology, our study also relates to Na, K and Ca However, the samples analyzed by ICP-MS at the Pierre Sue Laboratory being ores, slags, basalts (volcanic pounces, lava and glasses), slags of refining and the major constitutive elements of these samples are Si, Ti, Al, Mg. These matrix elements are thus the subject of the study.
The bibliographical study made possible to describe the spectroscopic interferences and to define the parameters highlighting the origin of the interference and thus controllable by the experimenter. The listed parameters are: nebuliser flow, observation position in plasma, elements masses and electronegativity. Initially, we were confronted with difficulties related to the reproducibility from day to day. Part of the study was devoted to determine the parameters (mainly, values of the tensions of the lenses) in front of imperatively remaining unchanged to have a reproducible signal during the measurement of the same solution. In the second time, we were interested in the influence of the mass of the element of matrix. In order to, we studied different masses elements but of the same chemical family. We compared the effects of the alkaline serie Na, K and Cs on the one hand and of the halogens Cl and I on the other hand. From this way, one frees oneself to the maximum of the influence of energy of ionization and only the effect of space load is highlighted. Thus the signal of a solution containing Be, Al, V, Cr, Mn, Ni, Co, As, In, Ba, La, Ce, Re, Pb each one with a content of 10 ppb was recorded with and without 1000 ppm matrix element for several radial distances. The evolution of the degree of extinction of the signal according to the radial distance for each matrix was given.
Figure 2 shows that the Al signal is uplifted by sodium (degree of extinction > 1) whereas it is slightly extinguished by K and Cs. It is noticed that each time the signal is practically constant with the radial distance. Thus, we can eliminate an origin from the exaltation related to the diffusion in plasma. The signal modification is related to the entry of the ions in the interface. While entering the interface, all the ions have same speed but the heaviest ions have a more important kinetic energy and thus undergo less than the light ions the effect of repulsion between positive ions in the beam of small diameter. It is what explains why the ions of high mass are transmitted better. In the case of Pb, figure 3 presents a different influence from the alkaline elements. Indeed the influence of their mass seems negligible on this heavy ion. This time, the signal varies with the radial distance for Na and Cs. There is a displacement of the zone of formation of the ions, according to the alkaline one, this which will involve a different diffusion in plasma. This displacement of ion-atom equilibrium and rather related to the potential of Pb ionization raised in front of that of alkaline and Al.
1. Investigation of plasma-related matrix effects in inductively coupled plasma-atomic emission spectrometry caused by matrices with low second ionization potentials - identification of the secondary factor.
G.C.-Y. Chan and G.M. Hieftje, Spectrochimica Acta Part B, 61(6) (2006) 642.
2. Effect of concomitant elements on the distribution of ions in inductively coupled plasma mass spectrometry. Part 1 Elemental ions.
M.M. Fraser and D. Beauchemin, Spectrochimica Acta Part B, 55 (2000) 1705.
3. The effect of easily ionisable concomitant elements on non spectroscopic interferences in inductively coupled plasma mass spectrometry, D.C. Gregoire, Spectrochimica Acta Part B. 42(6) (1987) 895.
4. Effects of ionisable elements on the liquid sampling - atmospheric pressure glow discharge
J.L. Venzie and R.K. Marcus, Spectrochimica Acta Part B, 61 (2006) 715.
5. Effect of sample matrix on the fundamental properties of the inductively coupled plasma.
S.A. Lehn, K.A. Warner, M. Huang and G.M. Hieftje Spectrochimica Acta Part B. 58(10) (2003) 1785.