

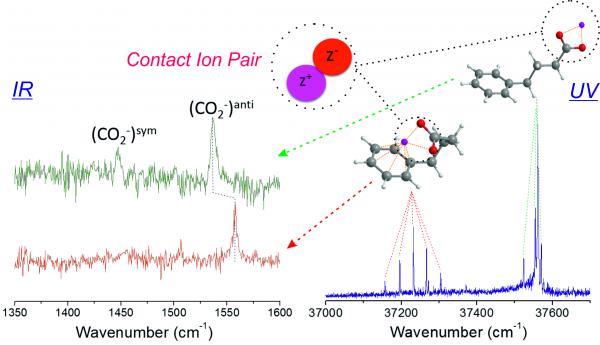
IR and UV spectroscopy reveal the molecular folding resulting from the competition between two non-covalent intramolecular interactions: cation-anion and cation-pi.
Ion Pairs
Ion pairs are ubiquitous in Nature, from sea water and aerosols, to living organisms. Being the very first step of crystallization of ionic species and influencing the properties of ion-concentrated solutions or ionic liquids, they also play a key role in countless applications. Although they are met in many areas of Physics, Chemistry and Biology, their characterization is complicated by the co-existence of several types of pairs and their elusive nature in solution.
In this context, this project aims at documenting net neutral ion pairs by investigating them in the gas phase using IR and UV laser spectroscopic techniques which enable us to characterize each type of ion pair individually. This experimental approach is combined with theoretical approaches aiming at identifiying the ion pairs formed in the gas phase, and extrapolating their vibrational signature in solution. Several questions are investigated by this project:
- the gas phase structure of model ion pairs.
- the application of gas phase results to ion pair issues in solution.
- the investigation of Stark effects in isolated ion pairs.
This project is funded by:
- The French National Research Agency (2016-2020, JCJC grant ANR-16-CE29-0017)
- Synchrotron SOLEIL (2019-2020)
- The French National Computing Centers (2016-present, High Performance Computing resources, project A0050807524)
- The Japan Society for the Promotion of Science (2018, JSPS Summer Grant, J. Donon)
- Université Paris-Saclay (2014-2017, PhD grant of the Chemical Sciences Doctoral School of Université Paris-Saclay )
- LabEx PALM (2014-2018, Master student grants, ANR-10-LABX-0039-PALM)
Collaborations:
- Pr. David J. Aitken (Institut de Chimie Moléculaire et des Matériaux d'Orsay, Université Paris-Saclay)
- Dr. Denis Céolin (Soleil Synchrotron Facility, France)
- Pr. Masaaki Fujii, Dr. Shun-ichi Ishiuchi (Tokyo Institute of Technology, Japan)
Références:
J. Donon, PhD thesis Université Paris-Saclay (2020)
J. Donon, S. Habka, V. Vaquero-Vara, V. Brenner, M. Mons, E. Gloaguen J. Phys. Chem. Lett. (2019)
S. Habka, T. Very, J. Donon, V. Vaquero-Vara, B. Tardivel, F. Charnay-Pouget, M. Mons, D.J. Aitken, V. Brenner, E. Gloaguen, Phys. Chem. Chem. Phys. 21 12798 (2019)
S. Habka, PhD thesis Université Paris-Saclay (2017)
S. Habka, V. Brenner, M. Mons, E. Gloaguen J. Phys. Chem. Lett. (2016)
•  Laser-matter interaction › Physico-chemistry and Chemical-physics
Laser-matter interaction › Physico-chemistry and Chemical-physics
• Interactions, Dynamics and Lasers Laboratory (LIDYL) - CEA-CNRS and Paris Saclay University











