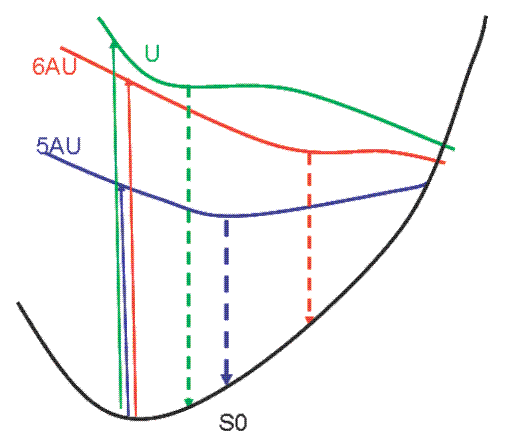
A detailed experimental and computational study of the absorption and fluorescence spectra of 5-aminouracil (5 AU) and 6-aminouracil (6 AU) in aqueous solution is reported. The lowest energy band of the steady-state absorption spectra of 5 AU is considerably red-shifted, noticeably less intense, and broader than its counterpart in uracil (U). On the contrary, the 6 AU lowest energy absorption peak is close in energy to that of U, but it is much narrower and the transition is much more intense. The emission properties of 5 AU, 6 AU, and U are also very different. Both amino-substituted compounds exhibit indeed a much larger Stokes shift as compared to U, and the emission band of 5 AU is much narrower than that of 6 AU. Those features are fully rationalized with the help of PCM/TD-PBE0 calculations in aqueous solution and MS CASPT2/CASSCF calculations in the gas phase. A stable minimum on the potential energy surface of the lowest energy bright state is found for 5 AU, both in the gas phase and in aqueous solution. For 6 AU a barrierless path leads to the conical intersection with the ground electronic state, but a nonplanar plateau region is predicted in aqueous solution, which is responsible for the very large Stokes shift. Some general considerations on the excited-state dynamics of uracil derivatives are also reported.
•  Physique et chimie pour le vivant et l’environnement
Physique et chimie pour le vivant et l’environnement
• Interactions, Dynamics and Lasers Laboratory (LIDYL) - CEA-CNRS and Paris Saclay University • Laboratoire Interactions, Dynamiques et Lasers (LIDYL) - CEA-CNRS et Université Paris Saclay • Service des Photons Atomes et Molécules
• Dynamics and Interactions in Condensed phase • Dynamique et Interactions en phase Condensée • Laboratoire Francis Perrin