•  Physique et chimie pour le vivant et l’environnement › Capteurs chimiques et biochimiques, diagnostic médical / Chemical and biochemical sensors, medical diagnosis
Physique et chimie pour le vivant et l’environnement › Capteurs chimiques et biochimiques, diagnostic médical / Chemical and biochemical sensors, medical diagnosis


In swimming pools, chlorine (Cl2) is used as a disinfectant to minimize the risk to users from microbial contaminants. But Cl2 is transformed in water into hypochlorous acid (HClO), which reacts with nitrogen compounds generated by human activity like saliva, sweat, urine and skin. This leads to the formation of several chloramines, such as monochloramine, dichloramine and the toxic nitrogen trichloride (NCl3). Because of its low solubility in water, NCl3 is mainly found in the air, and it provokes significant eye and respiratory irritations in swimmers and pool-attendants. Further, epidemiologic studies have recently shown that it can induce asthma, especially in children. The NCl3 content can vary within 10 min depending on the number of swimmers and the churning of water. In order to rapidly renew the pool atmosphere when needed, it is therefore important to be able to follow NCl3’s concentration. Up to now, the only method used to measure gaseous NCl3 is rather slow, since it is based on sampling with a cartridge of the atmosphere during 2 hours, followed by analysis in laboratory.
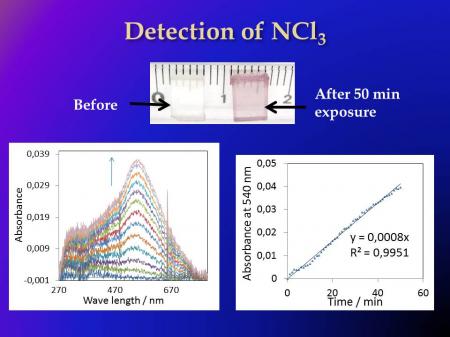
For the colorimetric NCl3 sensor, the laboratory has employed a strategy similar to that for formaldehyde detection. The well-known reaction of NCl3 with iodide ions in solution leads to the formation of the triiodide ion, I3-, which absorbs at 270 nm. In solution, this latter species can form with amylose the I3-/Amylose complex, which absorbs strongly in the visible at 540 nm. The key innovative idea was to transpose these reactions to nanopores doped with iodide and amylose. The developed sensor can detect NCl3 down to 5-200 ppb within 20 minutes in humid atmospheres (from 50% to 75% relative humidity) at ambient pool temperatures. The sensor is a transparent monolithic matrix of dimension 8X5X2 mm doped with sodium iodide and amylose. When it is exposed to NCl3, a color develops due to formation of the complex I3-/amylose; the color varies from light pink to dark violet depending on the NCl3 concentration.
With the system (Trichlor’Air) produced by ETHERA and commercialized by CIFEC, a sensor cartridge is placed in a fluidic chamber through which is passed during 20 min a constant flux of the air to be analysed. The intensity of the color, proportional to the NCl3 concentration, is measured at 580 nm with a reader unit equipped with a LED and a diode. With this sensor, it is now possible to also detect peaks of pollution in a pool, an ability not available with previous methods which give only average values over 2 hours.