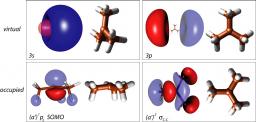
The excited state dynamics of the tert-butyl radical, t-C4H9, was investigated by femtosecond time-resolved photoionization and photoelectron spectroscopy. The experiments were supported by ab initio calculations. Tert-butyl radicals, generated by flash pyrolysis of azotert-butane, were excited into the A 2A1 (3s) state between 347 and 307 nm and the 3p band at 274 and 268 nm and ionized by 810 nm radiation, in a [1+2’] or [1+3’] process. Electronic structure calculations confirm that the two states are of s- and p-Rydberg character, respectively. The carbon framework becomes planar and thus ion-like in both states.
The photoelectron spectra are broad and seem to be mediated by accidental intermediate resonances in the probe step. All time-resolved photoelectron spectra can be described by a single decay time. For the A 2A1 state lifetimes between 180 and 69 fs were measured. Surprisingly a much longer lifetime of around 2 ps was found for the 3p state. In order to understand the decay dynamics, the potential energy was computed as a function of several important nuclear coordinates. A 1,2 H-atom shift to the iso-butyl radical seems not to be important for the excited state dynamic. Qualitative considerations indicate curve crossings between the ground state, the 3s state and a valence-state along the asymmetric C-C stretch coordinate that correlates to the dimethylcarbene + methyl product channel. The implications of the present study for earlier work on the nanosecond time scale are discussed.
•  Laser-matter interaction › Physico-chemistry and Chemical-physics
Laser-matter interaction › Physico-chemistry and Chemical-physics
• Service des Photons Atomes et Molécules • Service des Photons Atomes et Molécules