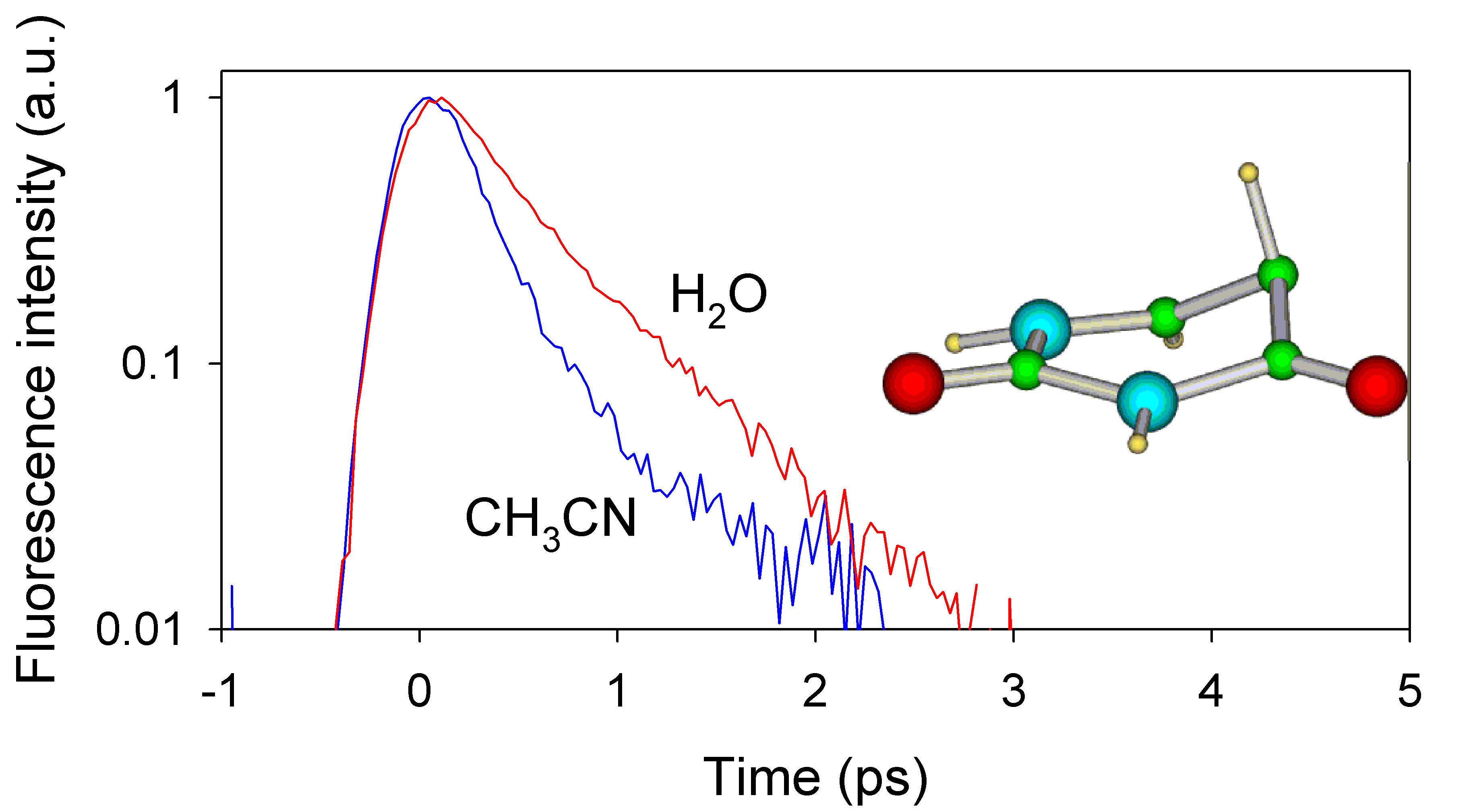
The excited state properties of uracil, thymine and four analogous uracil compounds have been studied in acetonitrile by steady-state and time-resolved spectroscopy. The excited state lifetimes were measured using femtosecond UV fluorescence upconversion. The excited state lifetimes of uracil and its 1- and 3-methyl substituted derivatives are well described by one ultrafast (100 fs) component. Five substituted compounds show a more complex behavior, exhibiting longer excited state lifetimes and bi-exponential fluorescence decays. These longer decays are substantially faster in acetonitrile than in aqueous solution showing that the excited state deactivation mechanism is in part governed by the solvent.
•  Physics and chemistry for life sciences and the environment › Physics and life
Physics and chemistry for life sciences and the environment › Physics and life
• Service des Photons Atomes et Molécules • Service des Photons Atomes et Molécules