Relaxation studies in the presence of off-resonance rf irradiation
Aim: The classical way to see relaxation in liquids
is strongly associated to Bloch’s equations, characterized by a longitudinal T1
and a transverse T2. This obviously needs to be extended in a first
step to dipolar cross relaxation as explored by NOESY or ROESY experiments and
in a further step to any mechanism involving cross-correlation induced
relaxation. What we have studied for a while has led to the definition of a
general unified view which links longitudinal relaxation along the static
magnetic field direction and transverse one performed in the presence of rf irradiation.
Principle: The key point consists in applying
the rf irradiation of amplitude
w1 not on-resonance as usual but off-resonance at
a distance D in Hz from the
spectrum centre. In the frame rotating at the rf field frequency this defines an effective field
and we can study the relaxation along this field. This effective field makes an
angle q=atan(w1/D) with the static magnetic field
direction and its amplitude W is defined by W2= w12+D2.
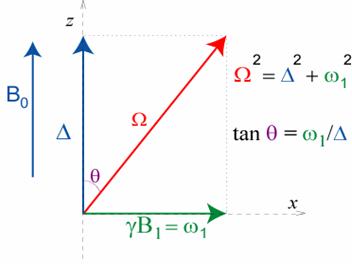
Definition and amplitude of the effective field. The magnetic fields are
drawn in a frame rotating at the rf
frequency.
Advantages:
- By varying the angle q, i.e. D and w1, the direction of the effective field
continuously varies from the longitudinal case to the transverse one.
Obviously a large number of relaxation rates correlated by the q and W values can be experimentally
determined. Depending on the relaxation mechanisms considered two or more
dynamic pieces of information can be obtained. Usually only two, the
longitudinal and transverse relaxation rates, which correspond to q=0 and q = 90°. The precision on their
determination is nevertheless increased since it benefits from the
absolute relation between the measured rates and these two limit values
which is dependent in a perfectible predictable way of the angle q.
- By increasing the offset D it becomes possible to probe
molecular dynamics at a scale on the order of 1/W which means about two orders
of magnitude larger than what can be achieved by on-resonance rf irradiation. This allows
the exploration of very slow overall dynamics or fast chemical exchange
appearing in the micro to millisecond time scale, and in fact using this
approach we had reported the fastest chemical exchange rate or the
determination of the whole chemical exchange spectral density function.
- In term of implementation the
two drawbacks of on-resonance rf
irradiation studies disappear. Indeed a large offset D induces that chemical shift
variation along the spectrum becomes negligible relative to this offset.
However in term of in-phase coherence transfer due to scalar couplings J (Homonuclear Hartmann-Hahn transfer or TOCSY) the
difference of chemical shifts between two protons remains non small
relative to J, preventing this type of transfer. This remark has allowed
us to propose the most reliable pulse sequence to study cross-relaxation
between protons in small to medium size molecule, i.e. off-resonance ROESY
or Tilted ROESY in the Bruker’s nomenclature.
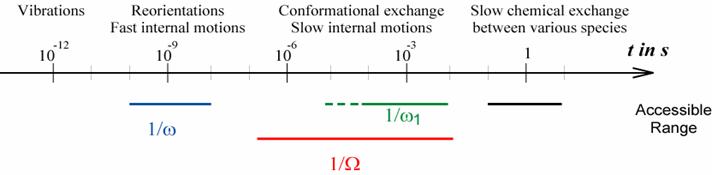
Domain of motions which can be studied by relaxation in the presence of off-resonance
rf irradiation.
Steady-state or transient measurements:
- Variation of the signal
intensities as a function of the mixing time is the straightforward
protocol for exploring relaxation in the presence of off-resonance rf irradiation. We have used
this principle for studying proton-proton dipolar cross-relaxation,
proton-proton dipolar cross-correlation, nitrogen relaxation in proteins,
proton or nitrogen chemical exchange, relaxation of paramagnetic proteins,…
- The measurement of the
steady-state magnetization in the presence of off-resonance rf irradiation is an other
protocol which directly gives access to dynamic parameters providing that
a model of dynamics is chosen. This constraint is counterbalanced by the
fact that thus each measurement provides dynamical information against
which the model can be tested. This was a useful approach for fast
determination of proton correlation times or of chemical exchange in
proteins.