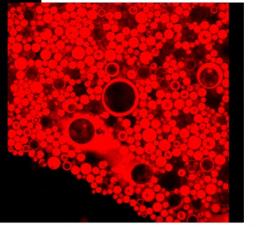
Emulsions are still delicate materials to make and their behavior is hard to predict since they are out-of-equilibrium objects. Multiple emulsions are even more complex to master since they usually require two surfactants to accommodate adverse curvatures. Consequently, they are difficult to make and poorly stable though the potential demand is high for delivery in particular if they can be stimulable. This is precisely that we recently achieved by using purposely designed copolymers of poly(styrene) and poly(madame) where the latter monomer is thermosensitive and charged as a function of pH. It was already demonstrated by us that the nature of emulsions from oil in water to water in oil, can be inverted by the two stimuli (T, pH)[1]. We recently discovered that stable multiple emulsions form in a narrow region of pHs just inferior to values leading to inversion. This formation is correlated to a drop in surface tension (Fig.8)[2]. These multiple emulsions are formed in one step with a single polymeric surfactant and are stimulable by pH and T, leading to a controlled release of inner hydrophilic species.
These studies strive on a strong collaboration between ESPCI and LIONS also funded by RTRA Triangle de la Physique. Neutron scattering and reflectivity (LLB-ILL) are currently developped for measuring the polymer interfacial behaviors associated to various kinds of emulsions
[1] F. Marchal, A. Roudot, N. Pantoustier, P. Perrin, J. Daillant and P. Guenoun, J. Phys. Chem. B, 111, 13151, (2007)
[2] L. Besnard, F. Marchal , J. F. Paredes , J. Daillant , N. Pantoustier , P. Perrin , P. Guenoun, Adv. Materials, 25, 2844, (2013)
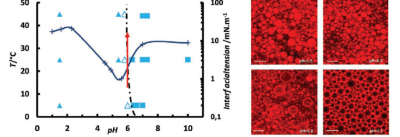
Left : surface tension and phase diagram in the plane pH, temperature. Full triangles denote o/w emulsions, open triangles multiple emulsions and squares w/o emulsions. Right : Corresponding confocal images of emulsions at pH 1, 4, 6 and 7 from left to right and top to bottom respectively. Each scale bar is 20 microns.
• › Interfaces, fluides complexes et microfluidique  Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects Synthesis and characterization of nano-objects
Synthèse et caractérisation des nano-objets / Synthesis and characterization of nano-objects Synthesis and characterization of nano-objects
• UMR 3299 - Service Interdisciplinaire sur les Systèmes Moléculaires et les Matériaux • Service Interdisciplinaire sur les Systèmes Moléculaires et les Matériaux
• Laboratoire Interdisciplinaire sur l'Organisation Nanométrique et Supramoléculaire (LIONS) • Interdisciplinary Laboratory on Nanoscale and Supramolecular Organization