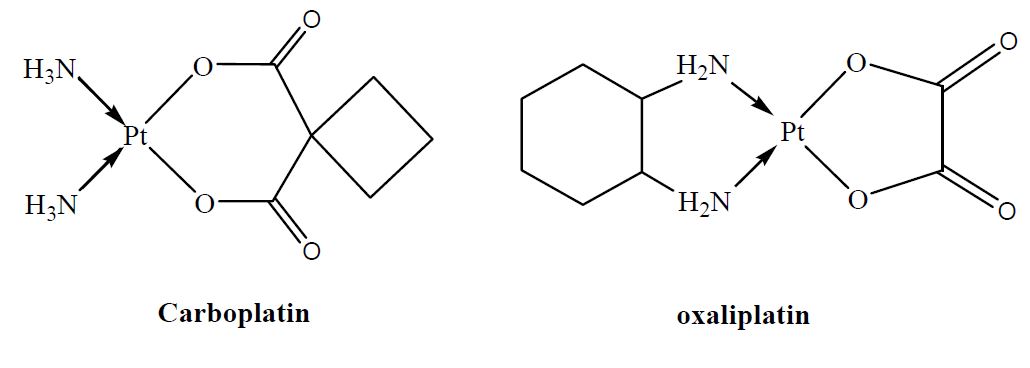
Cisplatin (cis-diamminedichloroplatinum(II)) is a widely used anticancer drug that has been particularly successful in treating small cell lung, ovarian, or neck tumors. However, cisplatin displays severe toxic side effects, and often during therapy resistance is developed. This has motivated motivated searches for structurally and/or functionally analogous alternatives. Unfortunately, finding analogous compounds that outperform cisplatin has proven to be difficult, and a better understanding at the molecular level of the interactions taking place between the Pt and the nucleobase moieties, is thought to be helpful in establishing a rational strategy to design new platinum-based analogues. In this context, operating in the gas phase makes it possible to overcome any solvent or counter-ion effect, and thus allows accessing the intrinsic reactivity of metal cations. Presently, we used the combination of several experimental approaches to theoretical calculations to characterize the interactions established between 3 platinum-based agents, namely cisplatin, oxaliplatin and carboplatin, with several DNA building blocks.
Experimentally, we generally use mass spectrometry together with InfraRed Multiple Photon dissociation (IRMPD) spectroscopy and/or ion mobility to obtain a detailed description of the complexes generated in the gas phase. In the present study, complexes were generated in the gas phase by electrospray ionization. IRMPD spectra in the mid-infrared region (900 cm-1-1900 cm-1) were recorded at the CLIO free electron laser facility (Centre Laser Infrarouge d'Orsay). Additional experiments in the 2600-3800 cm-1 energy range were also carried out through the use of an OPO/OPA laser. Theoretical calculations were carried out at B3LYP/6-311G(d,p) level. In order to describe the metallic centre, we will use the Los Alamos effective core potential (ECP) in combination with the LACV3P** basis set.










