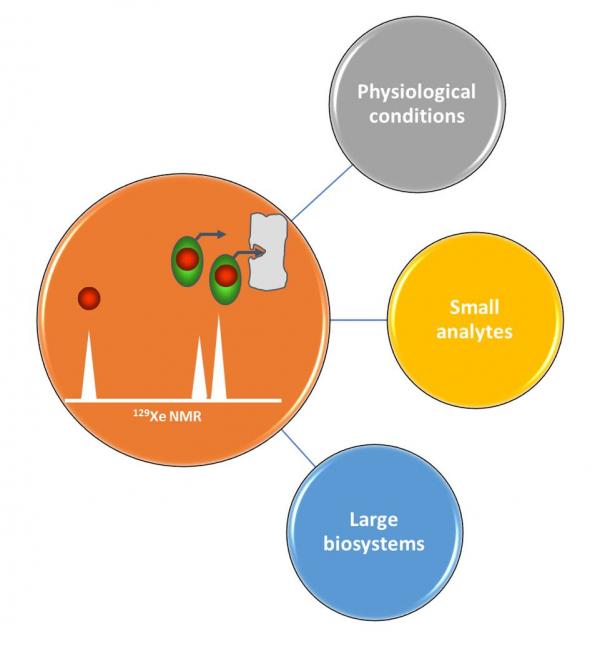
A long-term collaboration exists with mainly two chemistry labs: ENS Lyon and CEA/IBITEC-S to conceive and study xenon 129Xe NMR-based biosensors. Several molecular systems for which xenon develops a particular affinity and that give the noble gas a specific resonance frequency well resolved from the bulk signal, have been chemically decorated with biological ligands
Biological targets
- pH
Cancer, apoptosis
Chem. Eur. J. 16 (2010) 12941
- Metal ions
Cardiac diseases, diabetes, cancer, neurodegeneration
Angew. Chem. 51 (2012) 4100 DOI: 10.1002/anie.201109194
Anal. Chem. 86 (2014) 1783 DOI: 10.1021/ac403669p
- H2O2
Oxydative stress
Angew. Chem. 53 (2014) 9837
- DNA
- Cell surface receptors
Bioorg. Med. Chem. 19 (2011) 4135 DOI: 10.1016/j.bmc.2011.05.002
- Recombinant proteins
Chem. Comm. 51 (2015) 11482 DOI: 10.1039/C5CC04721H
- Cancer cells
Bioorg. Med. Chem. 25 (2017) 6653 DOI: 10.1016/j.bmc.2017.11.002
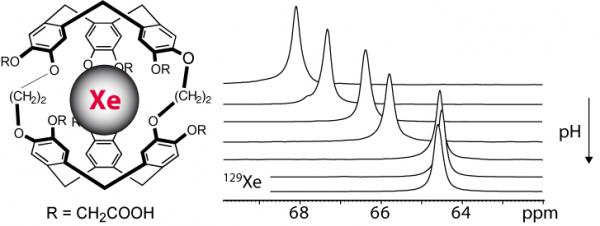
Measurement of pH
Local modification of pH is a key parameter in different pathologies such as cancers or biological processes such as apoptosis. We show that the sensitivity of xenon to its local environment and the presence of ionic groups on the host molecule can lead to interesting properties. For a first water-soluble cryptophane derivative, we show that a precise monitoring of the local pH can be performed. For a second cryptophane, the presence of ionic groups close to the cryptophane cavity modifies the xenon binding constant and in–out exchange rate. The latter allows the tuning of physical properties of xenon–cryptophane interactions without resorting to a change of the cavity size. These results open new perspectives on the influence of chemical modifications of cryptophanes for optimizing the biosensor properties.
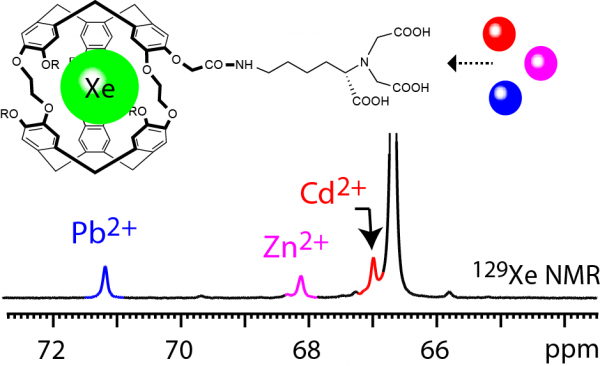
Detection of metal ions
Metal ions are involved in differents pathologies such as cardiac diseases, diabetes, cancer, neurodegeneration. Here, an approach for sensitive magnetic resonance detection of metal cations is proposed. Combining the use of hyperpolarized 129Xe NMR and of a cage-molecule functionalized by a ligand able to chelate different cations, we show that simultaneous detection of lead, zinc, and cadmium ions at nanomolar concentration is possible in short time, thanks to fast MRI sequences based on the HyperCEST scheme.
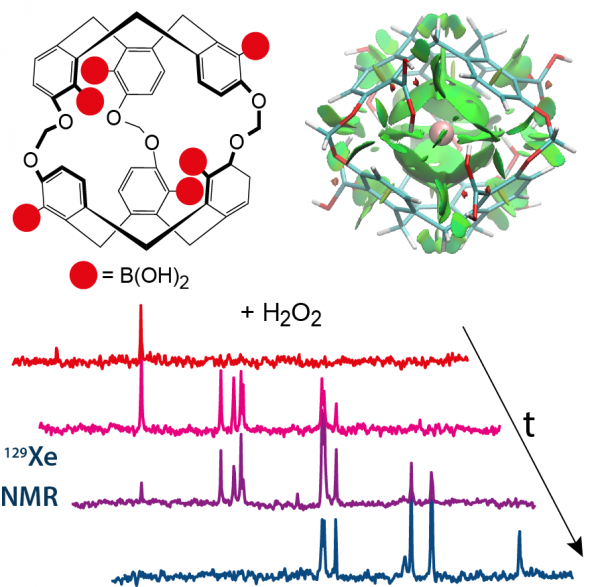
Detection of hydrogen peroxyde
Hydrogen peroxyde induces oxydative stress on cells, involved in the development of pathologies such as cancers, heart failures, Parkinson's disease. We propose here a probe able to detect H202 using 129Xe NMR.
Understanding the nature of host–guest interactions in supramolecular complexes involving heavy atoms is a difficult task. Described herein is a robust simulation method applied to complexes between xenon and members of a cryptophane family. The calculated chemical shift of xenon caged in a H2O2 probe, as modeled by quantum chemistry with complementary-orbital, topological, and energy-decomposition analyses, is in excellent agreement with that observed in hyperpolarized 129Xe NMR spectra. This approach can be extended to other van der Waals complexes involving heavy atoms.
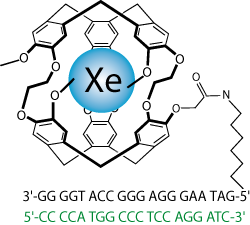
Detection of a specific DNA strand
A xenon host composed of a cryptophane structure with a DNA strand serves to detect its complementary strand in the micromolar range through laser-polarized 129Xe NMR spectroscopy.
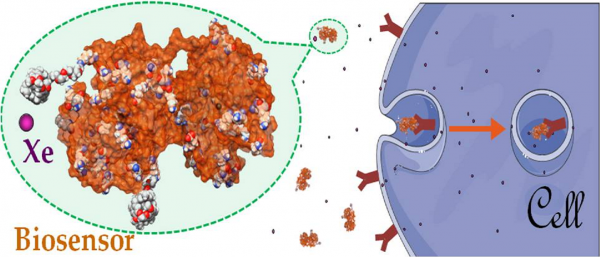
Detection of a cell surface receptor: the transferrin case
For detection of biological events in vitro, sensors using hyperpolarized 129Xe NMR can become a powerful tool, provided the approach can bridge the gap in sensitivity. Here we propose constructs based on the non-selective grafting of cryptophane precursors on holo-transferrin. This biological system was chosen because there are many receptors on the cell surface, and endocytosis further increases this density. The study of these biosensors with K562 cell suspensions via fluorescence microscopy and 129Xe NMR indicates a strong interaction, as well as interesting features such as the capacity of xenon to enter the cryptophane even when the biosensor is endocytosed, while keeping a high level of polarization. Despite a lack of specificity for transferrin receptors, undoubtedly due to the hydrophobic character of the cryptophane moiety that attracts the biosensor into the cell membrane, these biosensors allow the first in-cell probing of biological events using hyperpolarized xenon.
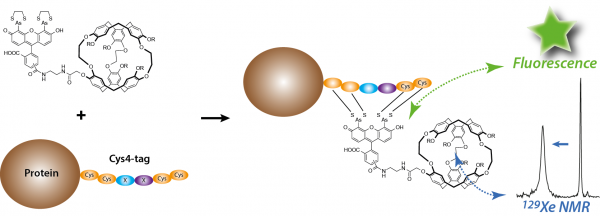
Recombinant proteins
Recombinant proteins bearing a tag are crucial tools for assessing protein location or function. Small tags such as Cys4 tag (tetracysteine; Cys–Cys–X–X–Cys–Cys) are less likely disrupt protein function in the living cell than green fluorescent protein. Herein we report the first example of the design and synthesis of a dual fluorescence and hyperpolarized 129Xe NMR-based sensor of Cys4-tagged proteins. This sensor becomes fluorescent when bound to such Cys4-tagged peptides, and the 129Xe NMR spectrum exhibits a specific signal, characteristic of the biosensor-peptide association
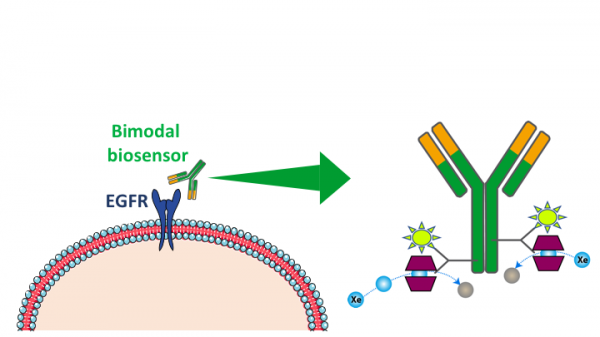
Cancer cells
Epidermal Growth Factor Receptors (EGFR) are overexpressed in many cancer cells such as the one involved in Non-Small Cell Lung Cancer (NSCLC). In this study in collaboration with IGR (Institut Gustave Roussy) we design a bimodal biosensor for fluorescence and hyperpolarized 129Xe NMR. It consists of 3 parts: 1) an antibody with a high affinity for EGFR, 2) a fluorophore and 3) a cryptophane. We have demonstrated on cell cultures that the biosensor reach EGFR receptors and we have been able to detect it at sub-nanomolar concentration, using fluorescence and 129Xe NMR sequences based on the HyperCEST effect.
•  Physique et chimie pour le vivant et l’environnement › Capteurs chimiques et biochimiques, diagnostic médical / Chemical and biochemical sensors, medical diagnosis
Physique et chimie pour le vivant et l’environnement › Capteurs chimiques et biochimiques, diagnostic médical / Chemical and biochemical sensors, medical diagnosis
• Laboratoire Structure et Dynamique par Résonance Magnétique (LSDRM)