|
Foldamers and H-bonding : the TUNIFOLDS project
The aim of this project to conceive new foldamer building blocks whose flexibility can be finely tuned thanks to the presence of an aliphatic thioether function, and to explore the ability of these building blocks to induce new folding topologies in backbones which do not normally fold, or to promote alternative folding architectures in short oligomers. The originality of the project, which unites organic chemists, gas phase and condensed spectroscopists and theoretical chemists, lies in the unique combination of (a) the exploitation of the specific properties of sulfur for the fine tuning of non-covalent interactions in foldamer manifolds; (b) the characterization of foldamer model conformations using conformer-selective spectroscopic gas phase techniques, which is still highly unusual in the foldamer field, as well as vibrational circular dichroism in solution; and (c) the role of theory for guidance during the selection of appropriate building blocks.
Effects of sulfoxide and sulfone sidechain-backbone hydrogen bonding on local conformations in peptide models
D.Y. Liu, S. Robin, E. Gloaguen, V. Brenner, M. Mons, D.J. Aitken ;
Chemical Communications
2024, in press
Length-Dependent Transition from Extended to Folded Shapes in Short Oligomers
of an Azetidine-Based
a-Amino Acid: The Critical Role of NH - N H-Bonds conformation in a flexible single-residue model
D.Y. Liu, J.-X. Bardaud, Z. Imani, S. Robin, E. Gloaguen, V. Brenner, D.J. Aitken, M. Mons ;
Molecules
2023, 28, 5048
Non-covalent interactions reveal the protein chain d conformation in a flexible single-residue model
Z. Imani, V.R. Mundlapati, V. Brenner, E. Gloaguen, K. Le Barbu-Debus, A. Zehnacker-Rentien, S. Robin, D.J. Aitken, M. Mons ;
Chemical Communications
2023, 59, 1161-1164
Selenium in Proteins: Conformational Changes Induced by Se Substitution on Methionine, as Studied in Isolated Model Peptides by Optical Spectroscopy and Quantum Chemistry
G. Goldsztejn, V.R. Mundlapati, V. Brenner, E. Gloaguen and M. Mons ;
Molecules
2022, 27, 3163
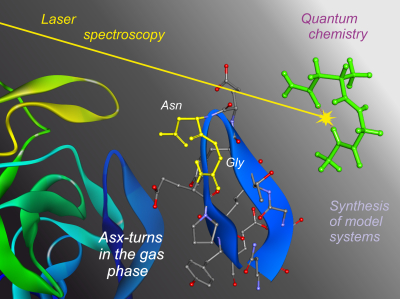
Characterization of Asx-turn types and their connate relationship with
b-turns
V.C. D'mello, G. Goldsztejn, V.R. Mundlapati, V. Brenner, E. Gloaguen, F. Charnay-Pouget, D.J. Aitken and M. Mons ;
Chemistry, A European Journal
2022, 28 , e202104328
N-H - - - X interactions stabilize intra-residue C5 hydrogen bonded conformations in heterocyclic a-amino acid derivatives
V.R. Mundlapati, Z. Imani, V.C. D'mello, V. Brenner, E. Gloaguen, J.-P. Baltaze, S. Robin, M. Mons, D.J. Aitken ;
Chemical Science
2021, 12, 14826-14832
A theoretical and experimental case study of the hydrogen bonding predilection of S-methylcysteine
V.R. Mundlapati, Z. Imani, G. Goldsztejn, E. Gloaguen, V. Brenner, K. Le Barbu-Debus, A. Zehnacker, J.-P. Baltaze, S. Robin, M. Mons, D.J. Aitken ;
Amino Acids
2021, 53, 621-633
Conformation control through concurrent N-H - - - S and N-H - - - O=C hydrogen bonding and hyperconjugation effects
Z. Imani, V.R. Mundlapati, G. Goldsztejn, V. Brenner, E. Gloaguen, R. Guillot, J.-P. Baltaze, K. Le Barbu-Debus, S. Robin, A. Zehnacker, M. Mons, D.J. Aitken ;
Chemical Science
2020, 11, 9191-9197
Intrinsic folding of the cysteine residue: competition between compact and extended forms mediated by the -SH group
G. Goldsztejn, V.R. Mundlapati, J. Donon, V.Brenner, E. Gloaguen, M. Mons, C. Cabezas, I. Leon and J.L. Alonso ;
Phys. Chem. Chem. Phys.
2020, 22, 20284-20294 ; 2020 PCCP HOT Article
An Intraresidue H-bonding motif in selenocysteine and cysteine, revealed by gas phase laser spectroscopy and quantum chemistry calculations
G. Goldsztejn, V. R. Mundlapati, J. Donon, B. Tardivel, E. Gloaguen, V. Brenner and M. Mons ;
Phys. Chem. Chem. Phys.
2020, 22, 20409-20420 ; 2020 PCCP HOT Article
Rationalizing the diversity of amide-amide H-bonding in peptides using the Natural Bond Orbital method
V. Brenner, E. Gloaguen and M. Mons ;
Phys. Chem. Chem. Phys.
2019, 21, 24601-24619
Vibrational circular dichroism as a probe of solid-state organisation of derivatives of cyclic b-amino acids: cis- and trans-2-aminocyclobutane-1-carboxylic acid
V. Declerck, A. Perez-Mellor, R. Guillot, D.J. Aitken, M. Mons, A. Zehnacker ;
Chirality
2019, 31, 547-560
Intrinsic folding proclivities in cyclic b-peptide building blocks: configuration and heteroatom effects analyzed
by gas phase conformer-selective spectroscopy and quantum chemistry
M. Alauddin, E. Gloaguen, V. Brenner, B. Tardivel. M. Mons, A. Zehnacker-Rentien, V. Declerck, D.J. Aitken
;
Chemistry, a European Journal
2015, 21, 16479-16493
|
|
Model peptide
chains
The interest of studies of model peptides
stems from the possibility to collect precise spectroscopic data,
which can be used as experimental reference to validate the theoretical models
describing the small peptides, a necessary step for protein modelling.
The small peptides studied, protected at their ends, can be considered as
mimicking a short peptide segment and therefore enable us to tackle
fundamental issues like the competition between the several secondary
structures of proteins, namely b-turns, b-strands, 27 ribbons or 310 and a helices.
Most recently, our interest focused on hydrates
of models of protein chains, on polar protein residues, such as His, Asn or Asp,
which exhibits a great diversity of H-bonding interactions,
on exotic types of H-bonds, like those involving a S heteroatom, or on ion pairs modelling the interaction between a negatively charged residue and a metal cation.
In parallel, the photophysics of aromatic residues in these protein chain models was also targeted.
b-N-Heterocyclic Cyclobutane Carboximides:
Synthesis through a Tandem Base-Catalyzed Amidation/aza-Michael Addition Protocol and Facile Transformations
S. Barranco, F. Cuccu, D.Y. Liu, S. Robin, R. Guillot, F. Secci, V. Brenner, M. Mons, P. Caboni, D.J. Aitken, A. Frongia ;
Eur. J. Org. Chem.
2023, 26, e2023001
Excited States Computation of Models of Phenylalanine Protein Chains: TD-DFT and Composite CC2/TD-DFT Protocols
M. Lebel, T. Very, E. Gloaguen, B. Tardivel, M. Mons, V. Brenner ;
Int. J. Mol. Sci.
2022, 23, 621
Ion Pair Supramolecular Structure Identified by ATR-FTIR Spectroscopy and Simulations in Explicit Solvent
J. Donon, S. Habka, T. Very, F. Charnay-Pouget, M. Mons, D.J. Aitken, V. Brenner, E. Gloaguen ;
Chem Phys Chem
2021, 22, 2442-2455
Conformational Analysis by UV Spectroscopy: the Decisive Contribution of Environment-Induced Electronic Stark Effects
J. Donon, S. Habka, M. Mons, V. Brenner, E. Gloaguen ;
Chemical Science
2021, 12, 2803-2815
Review:
Neutral Peptides in the Gas Phase: Conformation and Aggregation
Issues
E. Gloaguen, M. Mons, K. Schwing and M. Gerhards ;
Chemical Reviews
2020, 120, 12490-12562
CC2 Benchmark for Models of Phenylalanine Protein Chains: 0-0 Transition Energies and IR Signatures of the pp* Excited State
MS. Dupuy, E. Gloaguen, B. Tardivel, M. Mons, V. Brenner ;
Journal of Chemical Theory and Computation
2020, 16, 601-611
Identification of ion pairs in solution by IR spectroscopy: crucial contributions of gas phase data and simulations
S. Habka, T. Very, J. Donon, V. Vaquero-Vara, B. Tardivel, F. Charnay-Pouget, M. Mons, D.J. Aitken, V. Brenner and E. Gloaguen ;
Phys. Chem. Chem. Phys.
2019, 21, 12798-12805
On the turn-inducing properties of asparagine: the structuring role of the amide side chain, from isolated model peptides to crystallized proteins
W.Y. Sohn, S. Habka, V. Vaquero-Vara, M. Géléoc, B. Tardivel, V. Brenner, E. Gloaguen and M. Mons ;
Phys. Chem. Chem. Phys.
2018, 20, 3411-3423
Unifying the microscopic picture of His-containing turns: from gas phase model peptides to crystallized proteins
W.Y. Sohn, S. Habka, E. Gloaguen and M. Mons ;
Phys. Chem. Chem. Phys.
2017, 19, 17128-17142
Correcting the record: the dimers and trimers of trans-N-methylacetamide
T. Forsting, H.C. Gottschalk, B. Hartwig, M. Mons, M.A. Suhm ;
Phys. Chem. Chem. Phys.
2017, 19, 10727-10737
Gas-Phase Spectroscopic Signatures of Carboxylate-Li+ Contact Ion Pairs: New Benchmarks For Characterizing Ion Pairing in Solution
S. Habka, V. Brenner, M. Mons, E. Gloaguen ;
J. Phys. Chem. Lett.
2016, 7, 1192-1197
Local NH - p interactions involving aromatic
residues of proteins: influence of backbone
conformation and pp* excitation on the p H-bond
strength, as revealed from studies of isolated
model peptides
W.Y. Sohn, E. Gloaguen, V. Brenner, M. Mons ;
Phys. Chem. Chem. Phys.
2016, 18, 29969-29978
Gas phase double resonance IR/UV spectroscopy of an alanine dipeptide analogue using a non-covalently bound UV-tag : observation of a folded peptide conformation in the Ac-Ala-NH2 toluene complex
E. Gloaguen, B. Tardivel. M. Mons ;
Structural Chemistry; Special issue O. Shishkin
2016,
27, 225-230
Intrinsic folding proclivities in cyclic b-peptide building blocks: configuration and heteroatom effects analyzed
by gas phase conformer-selective spectroscopy and quantum chemistry
M. Alauddin, E. Gloaguen, V. Brenner, B. Tardivel. M. Mons, A. Zehnacker-Rentien, V. Declerck, D.J. Aitken
;
Chemistry, a European Journal
2015, 21, 16479-16493
Intra-residue interactions in proteins: interplay between Serine or Cysteine side chains and backbone conformations, revealed by laser spectroscopy of isolated model peptides
M. Alauddin, H.S. Biswal, E. Gloaguen, M. Mons
;
Phys. Chem. Chem. Phys.
2015,
17, 2169-2178
Review:
Isolated Neutral Peptides
E. Gloaguen & M. Mons
;
Topics in Current Chemistry
2015, 364, 225-270
Volume dedicated to "Gas-phase IR Spectroscopy for the Structural Characterization of Biomolecules" (A.M Rijs & J. Oomens Ed.)
Secondary structures in Phe-containing isolated dipeptide chains: laser spectroscopy vs. quantum chemistry
Y. Loquais, E. Gloaguen, S. Habka, V. Vaquero-Vara, V. Brenner, B. Tardivel and M. Mons ;
J. Phys. Chem. A, Invited paper Special issue J.-M. Mestdagh
2015,
119, 5932-5941
Direct Spectroscopic Evidence of Hyperconjugation Unveils the Conformational Landscape of Hydrazides
E. Gloaguen, V. Brenner, M. Alauddin, B. Tardivel, M. Mons, A. Zehnacker-Rentien, V. Declerck, D. J. Aitken
;
Angew. Chem. Int. Ed.
2014,
53, 13756-13759
On the near UV photophysics of a phenylalanine residue: conformation-dependent pp* state deactivation revealed by laser spectroscopy of isolated neutral dipeptides
Y. Loquais, E. Gloaguen, M. Alauddin, V. Brenner, B. Tardivel, M. Mons
;
Phys. Chem. Chem. Phys.
2014, 16, 22192-22200
Non-radiative relaxation of UV photoexcited
phenylalanine residues: probing the role of
conical intersections by chemical substitution
M. Malis, Y. Loquais, E. Gloaguen, C. Jouvet, V. Brenner, M. Mons, I. Ljubic, N. Doslic
;
Phys. Chem. Chem. Phys.
2014,
16, 2285-2288
Unraveling Non Covalent Interactions within Flexible Biomolecules: from electron density topology to gas phase spectroscopy
R. Chaudret, B. de Courcy, J. Contreras-Garcia, E. Gloaguen, A. Zehnacker-Rentien, M. Mons, J.-P. Piquemal
;
Phys. Chem. Chem. Phys.
16 , 9876-9891
Spontaneous Formation of Hydrophobic Domains in Isolated Peptides
E. Gloaguen, Y. Loquais, J.A. Thomas, D.W. Pratt, M. Mons
;
J. Phys. Chem. B
2013,
117, 4945–4955
Unraveling the Mechanisms of Nonradiative Deactivation in Model Peptides Following Photoexcitation of a Phenylalanine Residue
M. Malis, Y. Loquais, E. Gloaguen, H.S. Biswal, F. Piuzzi, B. Tardivel, V. Brenner, M. Broquier, C. Jouvet, M. Mons, N. Doslic, and I. Ljubic;
J. Am. Chem. Soc.
2012,
134, 20340–20351
Far/Mid Infrared Signatures of Solvent-Solute Interactions in a Microhydrated Model Peptide Chain
M. Cirtog, A.M. Rijs, Y. Loquais, V.Brenner, B.Tardivel, E. Gloaguen and M. Mons
;
J. Phys. Chem. Lett.
2012,
3, 3307–3311
The strength of NH---S hydrogen bonds in methionine residues revealed by gas phase IR/UV spectroscopy
H.S. Biswal, E. Gloaguen, Y. Loquais, B. Tardivel and M. Mons
;
J. Phys. Chem. Lett.
2012,
3,
755-779
Compact folding of isolated four-residue neutral peptide chains: H-bonding patterns and entropy effects
R.J. Plowright, E. Gloaguen and M. Mons
;
Chem. Phys. Chem.
2011,
12,
1889-1899; K. Kleinermanns Special Issue
Structure of the Indole-Benzene Dimer Revisited
H. S. Biswal, E. Gloaguen, M. Mons, S. Bhattacharyya, P.R. Shirhatti, and S.Wategaonkar
;
J. Phys. Chem. A
2011, 115, 9485-9492 , D.W. Pratt Festschrift
Isolated Monohydrates of a Model Peptide Chain: Effect of a First
Water Molecule on the Secondary Structure of a Capped
Phenylalanine
H. S. Biswal, Y. Loquais, B. Tardivel, E. Gloaguen, M. Mons
;
J. Am. Chem. Soc.
2011,
133
, 3931-3942
Gas phase folding of a two-residue model peptide chain:
on the importance of an interplay between experiment and theory
E. Gloaguen, B. de Courcy, J. P. Piquemal, J. Pilmé, O. Parisel, R. Pollet, H. S. Biswal, F. Piuzzi, B. Tardivel, M. Broquier, M. Mons ;
J. Am. Chem. Soc.
2010,
120
,
11860–11863
Experimental and Theoretical Investigation of the Aromatic-Aromatic Interaction in
Isolated Capped Dipeptides
E. Gloaguen, H. Valdes, F. Pagliarulo, R. Pollet,
B. Tardivel, P. Hobza, F. Piuzzi, and M. Mons ;
J. Phys. Chem.
2010,
114
,
2973–2982; B. Soep Festschrift
Gas phase folding of a (Ala)4 neutral peptide chain:
spectroscopic evidence for the formation of a
b
-hairpin H-bonding pattern
E. Gloaguen,
R. Pollet, F. Piuzzi, B. Tardivel,
M. Mons ;
Phys. Chem. Chem. Phys.
2009, 11, 11385–11388
Intramolecular
chiral recognition in a jet-cooled short
peptide chain: gamma-turn helicity probed by a
neighbouring residue
E. Gloaguen,
F. Pagliarulo, W. Chin, V. Brenner, F. Piuzzi, B. Tardivel,
M. Mons ;
Phys. Chem. Chem. Phys.
2007, 9, 4491 ; Hot Paper,
Special Issue on the
spectroscopic probes of Molecular recognition
(M. Suhm, Ed.) Link to the journal
Spectroscopic
Evidences for the Formation of Helical
Structures in Gas Phase Short Peptide Chains
V.
Brenner, F. Piuzzi,
I. Dimicoli, B. Tardivel, and M. Mons
J. Phys. Chem. A 2007,111,
7347
Special Issue dedicated to R.E. Miller
Link to the journal
Chirality-controlled formation of b-turn secondary structures in
short peptide chains: gas phase experiment vs. quantum chemistry
V. Brenner, F. Piuzzi,
I. Dimicoli, B. Tardivel,
and M. Mons
Angew.
Chemie Int. Ed. 2007, 46
,
2463 Link to the journal
Review Article: Probing the competition
between secondary structures and
local preferences in gas phase isolated peptide backbones
Chin, W.; Piuzzi, F.;
Dimicoli, L.; Mons, M.
Phys. Chem.
Chem. Phys. Invited paper 2006, 8, 10033
Link to the journal
Gas phase formation of a 310-helix
in a three-residue peptide chain: Role of side chain-backbone
interactions as evidenced by IR-UV double resonance experiments
Chin, W.; Piuzzi, F.; Dognon,
J. P.; Dimicoli, L.; Tardivel, B.; Mons, M.
J. Am. Chem.
Soc. 2005, 127, 11900 Link to
the journal
Spectroscopic evidence for gas
phase formation of successive b-turns in a three-residue
peptide chain
Chin,
W.; Compagnon, I. ; Dognon, J.-P.;Canuel, C. ; Piuzzi, F.; Dimicoli, I. ; M.; von
Helden, G.; Meijer, G, and M. Mons
J. Am. Chem.
Soc. 2005, 127, 1388
Link to the journal
Secondary structures of Val-Phe and Val-Tyr(Me) peptide chains in the gas phase:
effect of the nature of the protecting groups
Chin,
W.; Dognon, J.-P.; Piuzzi, F.; Dimicoli, I.; Mons, M.
Mol. Phys. 2005, 103,
1579 Link to
the journal
Gas-phase models of g- turns: Effect of
side-chain/backbone interactions investigated by IR/UV spectroscopy and
quantum chemistry
Chin, W.; Piuzzi, F.;
Dognon, J. P.; Dimicoli, I.; Mons, M.
J. Chem.
Phys. 2005, 123,
084301 Link to
the journal
Secondary structures of short
peptide chains in the gas phase:
double resonance spectroscopy
of protected dipeptides
Chin,
W.; Dognon, J.-P.;Canuel, C. ;
Piuzzi, F.; Dimicoli, I. ; Mons, M.;Compagnon, I. ; von
Helden, G. and Meijer, G
J. Chem.
Phys. 2005, 122, 054317
Link
to the Journal
Intrinsic folding of small
peptide chains: Spectroscopic evidence for the formation of b-turns in the
gas phase
Chin,
W.; Dognon, J.-P.;Canuel, C. ;
Piuzzi, F.; Dimicoli, I. ; Mons, M.;Compagnon, I. ; von
Helden, G. and Meijer, G
J. Am. Chem. Soc. 2005, 127,
707 Link
to the journal
The gas-phase dipeptide analogue acetyl-phenylalanyl-amide:
A model for the study of side chain/backbone interactions in proteins
Chin, W.; Mons, M.; Dognon,
J. P.; Mirasol, R.; Chass, G.; Dimicoli, I.; Piuzzi, F.; Butz, P.;
Tardivel, B.; Compagnon, I.; von Helden, G.; Meijer, G.
J. Phys.
Chem. A 2005, 109, 5281 Link to
the journal
Characterization of the
conformational probability of N-acetyl-phenylalanyl-NH2 by RHF,
DFT, and MP2 computation and AIM analyses, confirmed by jet-cooled
infrared data
Chass, G. A.; Mirasol, R. S.; Setiadi, D.
H.; Tang, T. H.; Chin, W.; Mons, M.; Dimicoli,
I.; Dognon, J. P.; Viskolcz,
B.; Lovas, S.; Penke,
B.; Csizmadia, I. G.
J. Phys.
Chem. A 2005, 109, 5289 Link to
the journal
Competition between local
conformational preferences and secondary structures in gas-phase model tripeptides as revealed by laser spectroscopy and
theoretical chemistry
Chin,
W.; Mons, M.; Dognon, J.-P.; Piuzzi, F.; Tardivel, B. and Dimicoli, I.
Phys.
Chem. Chem. Phys. 2004, 6, 2700 Link to the journal
Other molecules in supersonic expansions
Spectroscopy in supersonic expansions provides precise spectroscopic data, which enable us to improve theoretical models and molecular simulation
Rovibrational laser jet-cooled spectroscopy of SF6-rare gas complexes in the ?3 region of SF6
P. Asselin, A.C. Turner, L. Bruel, V. Brenner, M.-A. Gaveau, M. Mons
Phys. Chem. Chem. Phys., 2018, 20, 28105-28113
Conformational landscape of the SF6 dimer as revealed by high resolution infrared spectroscopy and complexation with rare gas atoms
P. Asselin, A. Potapov, A.C. Turner, V. Boudon, L. Bruel, M.-A. Gaveau, M. Mons
Phys. Chem. Chem. Phys., 2017, 19, 17224-17232
DNA Bases
There is a dual motivation for these studies:
·
To collect precise spectroscopic reference
data to validate the calculations
carried out on these biological systems
·
To understand the relaxation dynamics of the
excited states of DNA bases; these electronic states are indeed
responsible of DNA absorption in
the UV and are potentially a doorway to irreversible damages to the
genetic code of living world.
Effective Strategy for Conformer-Selective Detection of Short-Lived Excited State Species: Application to the IR Spectroscopy of the N1H Keto Tautomer of Guanine
H. Asami, M.Tokugawa, Y. Masaki,S. Ishiuchi, E. Gloaguen, K. Seio, H. Saigusa, M.Fujii, M. Sekine and M. Mons
J. Phys. Chem. A, 2016, 120, 2179-2184
Ab Initio Kinetic Simulation of Gas-Phase Experiments: Tautomerization of Cytosine and Guanine
D. Kosenkov, D, Y. Kholod, L. Gorb, O. Shishkin, D.M. Hovorun, M. Mons, J. Leszczynski
J. Phys. Chem. B, 2009, 113, 6140-50
Near-UV Resonant Two-Photon Ionization
Spectroscopy of Gas Phase Guanine: Evidence for the Observation of Three
Rare Tautomers
M. Mons, I. Dimicoli, F. Piuzzi,
L. Gorb, J. Leszczynski,
J. Phys. Chem. A, 2006, 110, 10921
Time-resolved photoelectron and photoion fragmentation spectroscopy study of
9-methyladenine and its hydrates: a contribution to the understanding of
the ultrafast radiationless decay of excited
DNA bases
C. Canuel, M. Elhanine, M. Mons, F. Piuzzi, B. Tardivel and I.
Dimicoli
Phys. Chem.
Chem. Phys. 2006, 8, 3978 Link to the journal
Theoretical study of the ground and
excited states of Guanine, 7‑methyl Guanine, and 9‑methyl
Guanine: Comparison with experiment
J. Cerny, V. Spirko, M. Mons, P. Hobza, D. Nachtigallova
Phys. Chem. Chem. Phys. 2006, 6 , 3059 Link to the journal
Excited states dynamics of DNA
and RNA bases : Characterisation of a step-wise deactivation pathway in
the gas phase
Canuel, C.; Mons, M.;
Piuzzi, F.; Tardivel, B.; Dimicoli, I.; Elhanine, M.
J. Chem. Phys. 2005, 122,
074316 Link to the journal
Gas phase rotamers
of the nucleobase 9-methylguanine enol and its monohydrate: Optical spectroscopy and
quantum mechanical calculations
Chin, W.; Mons, M.; Piuzzi,
F.; Tardivel, B.; Dimicoli, I.; Gorb, L.; Leszczynski, J.
J. Phys.
Chem. A 2004, 108, 8237 Link to the journal
Tautomer contributions to the near UV spectrum of guanine: towards a
refined picture for the spectroscopy of purine
molecules
Chin,
W.; Mons, M.; Dimicoli, I.; Piuzzi, F.; Tardivel, B.; Elhanine, M.
Eur. Phys. J. D 2002, 20, 347
Link to the journal
Tautomerism of the DNA base guanine and its methylated
derivatives as studied by gas phase IR and UV spectroscopy
Mons,
M.; Dimicoli, I.; Piuzzi, F.; Tardivel, B.; Elhanine, M.
J. Phys. Chem.
A
2002, 106, 5088 Link to the journal
Ultraviolet spectroscopy and tautomerism of the DNA base guanine and its hydrate
formed in a supersonic jet
Piuzzi,
F.; Mons, M.; Dimicoli, I.; Tardivel, B.; Zhao, Q.
Chem. Phys. 2001, 270, 205 Link to the journal
A simple laser vaporization
source for thermally fragile molecules coupled to a supersonic expansion:
application to the spectroscopy of tryptophan
Piuzzi, F.; Dimicoli, I.;
Mons, M.; Tardivel, B.; Zhao, Q.
Chem. Phys. Lett. 2000, 320,
282 Link to the journal
|
|
Binding energy of a water molecule to several organic functional
groups
Measured at the end of the 90’s using a selective photofragmentation laser technique, these binding
energies are now considered as benchmark data, often cited in chemistry
and biochemistry studies
Benzene-water :
Quantum effects
in the threshold photoionization and energetics of the benzene-H2O and
benzene-D2O complexes: Experiment and simulation
Courty, A.; Mons, M.; Dimicoli,
N.; Piuzzi, F.; Gaigeot,
M. P.; Brenner, V.; de Pujo, P.; Millie, P.
J. Phys. Chem. A 1998,
102, 6590 Link to the
journal
Phenol-water:
Ionization, energetics, and geometry of the phenol-S
complexes (S = H2O, CH3OH,
and CH3OCH3)
Courty, A.; Mons, M.; Dimicoli,
I.; Piuzzi, F.; Brenner, V.; Millie, P.
J. Phys. Chem. A 1998,
102, 4890 Link to the journal
Amide-water:
Energetics of the gas phase hydrates of
trans-formanilide: A microscopic approach to
the hydration sites of the peptide bond
Mons,
M.; Dimicoli, I.; Tardivel, B.; Piuzzi, F.; Robertson, E. G.; Simons, J.
P.
J. Phys. Chem. A 2001,
105, 969 Link to the journal
Indole-water:
Site dependence of the binding
energy of water to indole: Microscopic approach
to the side chain hydration of tryptophan
Mons,
M.; Dimicoli, I.; Tardivel, B.; Piuzzi, F.; Brenner, V.; Millie, P.
J. Phys. Chem. A 1999,
103, 9958 Link to the
journal
Energetics of a model NH-p interaction: the gas phase benzene- NH3 complex
Mons,
M.; Dimicoli, I.; Tardivel, B.; Piuzzi, F.; Brenner, V.; Millie, P.
Phys. Chem. Chem. Phys. 2002,
4, 571 Link to
the journal
See also our review paper on
binding energies:
Gas phase hydrogen-bonded
complexes of aromatic molecules: photoionization
and energetics
Mons,
M.; Dimicoli, I.; Piuzzi, F.
Int. Rev. Phys.
Chem. 2002, 21, 101
Link to the journal
Structure of aromatic – rare gas clusters
Microscopic molecular solvation
was a very popular subject in the beginning of the 90’s, especially with
model solvents like rare gases atoms. Because of its apparent simplicity,
the benzene-argon system has been a case study and a benchmark for
numerous studies on aromatic molecule-rare gas systems.
Microsolvation of benzene by argon atoms : spectroscopy and isomers
Schmidt,
M., Mons, M.; Le Calvé, J.
Chem .
Phys. Lett. 1991, 177, 371 Link to the
journal
The second conformer of the benzene-Ar2 complex
Schmidt,
M., Mons, M., Le Calvé, J., Millié, P., Cossart-Magos, C.
Chem .
Phys. Lett.
1991, 183, 69
Link to the
journal
Resonant two-photon ionization spectra of the external vibrational modes of the chlorobenzene-,
phenol- and toluene-rare gas (Ne, Ar, Kr, Xe) van der Waals complexes
Mons, M.; Le Calvé, J. ; Piuzzi F., Dimicoli,
I.
J. Chem. Phys. 92 (1990) 2155
Link to the journal
Molecular photofragment characterisation
by laser spectroscopy
Our works on NO2 photodissociation
are amongst the first in this field and are still now often cited in the
literature, more than 15 years after publication, for both the specific
NO2 excited state physics and the original photofragment
detection technique used, which is sensitive to the angular correlation
between velocity and angular momentum .
Photodissociation of NO2. Internal energy distribution and anisotropies
in the fragments
Mons, M.; Dimicoli,
I.
Chem.
Phys. 130 (1989) 307 Link to the
journal
Angular correlations between photofragment
velocity and angular momentum measured by resonance enhanced multiphoton ionization detection
Mons, M.; Dimicoli,
I.
J. Chem.
Phys. 90 (1989) 4037
Link to the journal
State selective kinetic distribution
of photofragments
Mons,
M.; Dimicoli, I.
Chem.
Phys. Lett. 131
(1986) 298.
Link to the
journal
|